Triglycerides as a Factor in Residual ASCVD Risk
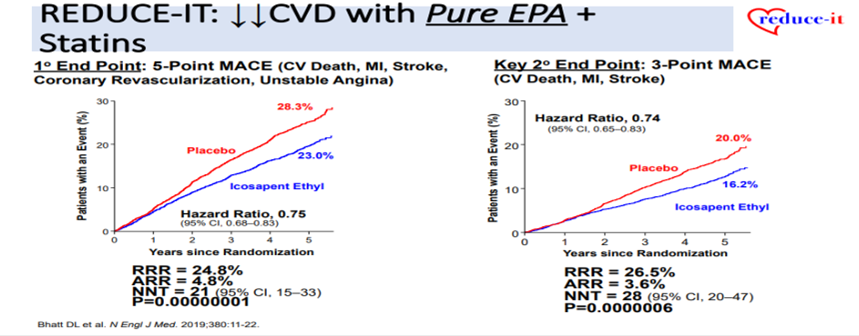
Brinton EA, talked about high TG and ASCVDin a session at American Association of Clinical Endocrinology (AACE) Annual Meeting 2022 on 13th May 2022. In PROVE IT-TIMI 22 trial, TG ≥150 mg/dL predicts higher CHD risk despite LDL-C<70 mg/dL on statin therapy. 50-80% of CVD still occurs despite statin monothearpy; much of that residual relates to high triglycerides (HTG). In MIRACL clinical trial, even normal to mildly-elevated TG associates with increased CVD Risk by 25-50%. AHA stated that a meal that contained up to 15 g of fat was associated with minimal (20%) increases in peak postprandial TG levels, whereas high-fat meals (ex. 50 g), including those served in popular fast-food restaurants, increased TG levels by at least 50% beyond fasting levels. Since TG-lowering medicines lower TG levels by only 20%-50%, the noise in non-fasting TG is minimal. According to IMPROVE-IT and FOURIER 2021 studies, Apo B predicts MI risk better than TG or Non-HDL-C. REDUCE-IT study showed that pure EPA plus statins significantly decreases the risk of CVD. In TG-Lowering Omega-3 CV Outcome Trials, low-dose EPA+DHA Mix (as diet-Supplement or therapy) showed no decrease in CVD. EPA increases heme oxygenase-1 expression, potentially increasing anti-oxidant/cytoprotective effects and preserves vascular endothelial function post il-6 exposure compared to DHA or AA. Proteomic analyses show that EPA downregulates pro-inflammatory proteinsand upregulates anti-inflammatory proteins but DHA does the opposite (like the pro-inflammatory IL-6 control). Pure EPA (IPE) is better than EPA + DHA Mix. Fibrates appeared to decrease CVD in patients with DM2/HTG/Low HDL-C but PROMINENT was negative. It seems appropriate to conclude from PROMINENT that fenofibrate is NOT useful for ASCVD prevention in HTG/Low HDLC patients. IPE is the only proven agent to decrease ASCVD in HTG/Low HDLC.
HTG patients have increased ASCVD risk by many mechanisms. Total TG, Non-HDLC (& apo B, if available) were used to assess the CV risk. Advanced lipid measurements are controversial; Apo B is very good but required extra test/money. Others (sd LDL, direct RLPC, VLDLP+IDLP, LDLP, etc.) can be inferred from TG & Non-HDLC and are likely not worth it, yet. To treat patients with HTG to lower ASCVD risk, always encourage diet and lifestyle first, manage diabetes (w/ pio, SGLT2i, GLP1-RA), obesity, high blood pressure. Statins are 1st-line in essentially all patients. If the TG is still >~135 mg/dL fasting (or >150 non-fasting), add IPE, if at all possible, Rx “DAW/brand only, generic IPE not indicated to decrease ASCVD or for TG <500.
Ethnic Diversity for Cardiovascular Disease Risks in South Asia

Hoang TD, presented the cardiovascular data of South Asians in a session at American Association of Clinical Endocrinology (AACE) Annual Meeting 2022 on 13th May 2022. The objective of the session was to review cardiovascular data available for South Asians and discuss treatment strategy for South Asian patients. South Asia (SA) is most densely populated regions on the planet and showed higher hospitalization and mortality rates from ASCVD compared with NHWs and other racial/ethnic minority groups. Indian men showed higher mortality than women CTA showed increase percentage stenosis and involvement of multiple vessels. Also, younger asians showed higher prevalence of DM and dyslipidemia compared with NHWs. SA more likely to have multivessel disease requiring CABG. Outcomes after PCI are generally similar in SA and NHWs. Some studies evaluating CV events and mortality after isolated CABG showed consistently poorer outcomes for SA. Statin is the mainstay therapy for hypercholesterolemia in South Asians. The suggested LDL-C goal is <100 mg/dL for high risk patients and < 70 mg/dL for very high risk. The LAI recommends an LDL-C goal < 50 mg/dL in all patients in secondary prevention or very high risk primary prevention but proposes an optional goal ≤30 mg/dL in extreme-risk patients (eg, coronary artery disease + familial hypercholesterolemia).
Rosuvastatin and Atorvastatin are well tolerated and effective in this population. Ezetimibe lowers plasma LDL-C by inhibiting the activity of NPC1L1 protein, resulting in reduced intestinal cholesterol absorption. IMPROVE-IT study exhibited that adding ezetimibe to simvastatin leads to 24% more LDL-C reduction. AIM-HIGH and HPS-2 THRIVE studies showed no incremental clinical benefit with Niacin. No South Asian studies have been conducted on the effects of niacin on CV outcomes. Statins primarily target LDL-C. Fibrates preferentially increase HDL-C, lower TG, and increase size of LDL-C particles particularly beneficial for South Asians.
In South Asians of US, evidence to date confirms the higher burden of CVD. It is recommended to use the QRISK3 calculator in clinical decisions to reduce CVD risk as early as childhood years and encourage healthy lifestyle in SA. Early recognition and treatment of CV risk factors is necessary to mitigate the CVD burden in SA. Imaging techniques (CAC) are significant for accurate risk stratification. Community strategies should be applied such as educate, promote health literacy and provide culturally competent health care.
Glycemic Monitoring and Use of Glucose Lowering Agents in Advanced CKD
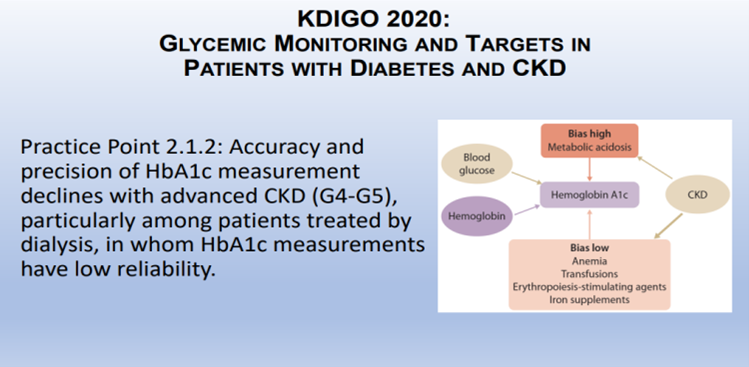
Galindo RJ, presented a session at American Association of Clinical Endocrinology (AACE) Annual Meeting 2022 on 13th May 2022. The session covered the current status of end stage kidney disease (ESKD) in diabetes mellitus (DM) patients, glycemic monitoring i.e. HbA1c, Glycated Albumin, Fructosamine, or CGM (goals) in advanced CKD, role of glucose lowering agents; orals, non-Insulin agents, Insulin, AIDs for advanced CKD. Insulin and glucose metabolism in CKD is altered and leads to increased risk of both hyperglycemia. hypoglycemia. According to KDIGO 2020, accuracy and precision of HbA1c measurement declines with advanced CKD (G4-G5), particularly among patients treated by dialysis in which HbA1c measurements have low reliability.
A glucose management indicator (GMI) derived from CGM data can be used to index glycemia for individuals in whom HbA1c is not concordant with directly measured blood glucose (BG) levels or clinical symptoms. Daily glycemic monitoring with CGM or SMBG may help to prevent hypoglycemia and improve glycemic control when antihyperglycemic therapies associated with risk of hypoglycemia are used. An individualized HbA1c target is recommended ranging from <6.5% to <8.0% in patients with diabetes and CKD not treated with dialysis. Linagliptin (no renal adjustment) (+/- basal insulin) or Sitagliptin is recommended for mild hyperglycemia. Repaglinide (short acting, ~4-5 hrs) or short-acting insulin can be given for meal related hyperglycemia. GLP1 agonists with proven CKD benefits are Dulaglutide, Semaglutide weekly and Liraglutide daily. As per AWARD study, Dulaglutide showed eGFR decline ≥40% or reduction in ESRD vs Insulin glargine. Insulin therapy is not shown to prevent progression of DKD. Starting with lower doses (0.25 units total daily dose), combine basal insulin with non-insulin agents, mixed insulin (70/30% or 75/25%) or BASAL – BOLUS should be the last option.
There is critical need for better glycemic monitoring in advanced CKD (1-2 finger sticks per day do not provide the full picture). CGM (eGMI) may overcome the limitation of HbA1c in this population and provide a more detailed assessment of glycemic excursion. Avoid over-insulization (higher risk of hypo and hyper crises). More studies and information is required on newer drugs DPP4 inhibitor, GLP1 agonist and devices in ESKD.
A Blind Spot: Weight Bias and Health Disparities in Obesity Care

Kahan S, presented a session on weight bias in health care at American Association of Clinical Endocrinology (AACE) Annual Meeting 2022 on 13th May 2022. Weight Bias is making (negative) assumptions or judgments (consciously or unconsciously) based on the appearance ofexcess weight. Persons with obesity are societally considered acceptable targets of stigma. As BMI increases patients receive less respect. 7 A’s” to address weight bias in healthcare are:
- Appreciate the complexity of obesity
- Awareness of implicit assumptions and beliefs
- Attention to what’s in our control
- Appreciate that shaming doesn’t motivate
- Support patients’ Autonomy
- Address consequences of stigma
- Advocate and Educate
Implicit anti-fat bias 2001 study stated that Implicit Association Test (IAT) indicated that more items were correctly classified when fat people was paired with negative attributes than with positive attribute. In attention to weight bias following points should be implemented:
- Provide wide-based, higher weight capacity chairs or consider specialized bariatric chairs, when possible
- Offer large (or thigh-sized) blood pressure cuffs and neutral waiting room literature
- Offer extra-large gowns and educate your staff about obesity and weight bias
Respondents who perceived judgment were significantly more likely to attempt weight loss. Among patients whose primary care providers (PCP) discussed weight loss, 20% achieved ≥10% weight loss if they did not perceive judgment by their PCP as compared to 14% who perceived judgment.
A RCT showed that a psychological intervention for weight bias internalization (WBI) produced short term reductions in weight self stigma in person with obesity. People should be advocated and educated about the complexity of obesity. Be an advocate in your practice hospital, community, and with your peers, advocate to policymakers and challenge weight bias in the public.
Weight bias refers to a wide range of discriminatory and harmful attitudes toward people deemed to be overweight. It can include beliefs such as that heavier people are usually unhealthy, having excess weight is easy to modify and is usually a person’s fault. Healthcare professionals are biased against people deemed as having overweight or obesity. It is also important to note that people with obesity can be healthy. Regardless of their weight or health practices, all people need access to quality, compassionate healthcare professionals who will not guilt, shame, or judge them.
Diagnosis and Investigation of Diabetes Insipidus

Kahric NJ, presented in nephrogenic diabetes insipidus in a session at American Association of Clinical Endocrinology (AACE) Annual Meeting 2022 on 13th May 2022. Water is the largest component of our body and since the major determinant of body water is AVP-regulated water excretion by the kidneys, therefore AVP is the most important hormone in the body. The types of diabetes insipidus are
- Central Diabetes Insipidus
- Hereditary Nephrogenic Diabetes Insipidus
- Acquired Nephrogenic Diabetes Insipidus
- Primary Polydipsia
- Gestational diabetes insipidus
- Osmoreceptor dysfunction
Central (neurogenic, pituitary, or neurohypophyseal) DI is characterized by decreased secretion of arginine vasopressin (AVP). Its prevalence is rare i.e. <1:25,000. Tumors associated with central DI are craniopharyngioma, germinoma (pinealoma), meningioma, lymphoma, metastatic (lung, breast). Mutations in the AVP gene is associated with hereditary central DI (autosomal dominant). Nephrogenic Diabetes Insipidus is even less common than neurogenic DI and it is clinically evident from birth. Mutations in the AVP V2 receptor gene is associated with hereditary nephrogenic DI. Primary polydipsia is most common cause of polyuria in Western countries. Dipsogenic polydipsia reset thirst threshold (mass lesions, granulomatous disease, idiopathic, aging) and psychogenic polydipsia increased fluid intake for reasons other than true thirst. The treatment of diabetes insipidus includes: Water, Antidiuretic agents (CDI) – AVP, desmopressin (DDAVP), chlorpropamide, carbamazepine and antiduresis enhancing agents (NDI) – Diuretics (thiazides, amiloride) and Indomethacin.
The general goals of treatment of all forms of DI are correction of any pre-existing water deficits and reduction of ongoing excessive urine losses. In adults, 50% of the deficit should be corrected in the first 24 h and the remainder over the next 24-48 hrs above the estimated urine output plus insensible looses. In children, correction of hypernatremia should not exceed 12 mmol/L/24h to minimize the risk of brain edema. The volumes replaced must include not only the calculated deficit, but also ongoing insensible and urine water losses; antidiuretic therapy is usually necessary to do this in DI.
Diabetes Technologies in 2022
Sadhu A, presented a session on automated algorithm in diabetes management at American Association of Clinical Endocrinology (AACE) Annual Meeting on 13th May, 2022. Machine Learning Algorithms in Automated Insulin Delivery (AID): Proportional Integral and Derivative (PID) – Used in various industries since 1940’s (automotive, manufacturing). Algorithm has three terms (Proportional, integral and derivative). It is a single input (CGM), single output (Insulin dose) systems. It has simpler dynamics, low computational burden. However, it uses only the current difference between measured (CGM) and desired blood glucose. Many additional modules added to predict blood glucose trends and to accommodate constraints. Constraints usually restrict the insulin dose computed by the algorithm and it is difficult to personalize the controller parameters.
AID Algorithms: Model Predictive Control (MPC) is based on advanced concepts in systems and control engineering. It has a higher computational burden caused by real-time optimization. It relies on availability of mathematical models describing glucose-insulin dynamic.
AID Machine Learning Algorithms: Fuzzy Logic- has a high cost of maintenance and revision of the rule base. As the numbers of rules are increased, conflict resolution algorithms are needed. Medical and programming expertise is needed for personalizing the system.
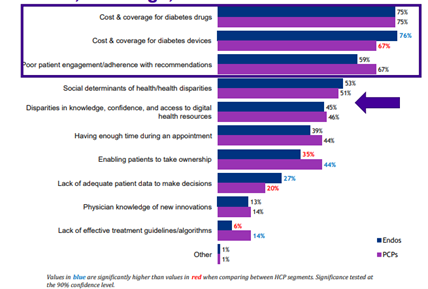
Three-quarters of HCPs selected cost and coverage for diabetes drugs as a barrier to improved outcomes. Similarly, cost and coverage for diabetes devices was also cited as a barrier by most HCPs, however, Endocrinologists are more likely to select this as a barrier than PCPs. Poor patient engagement/adherence with recommendations was also selected as a barrier by most HCPs. Over 40% of HCPs selected disparities in knowledge, confidence, and access to digital health resources as a barrier.
Adrenal Imaging Strategies – Which and When?
Blake M, presented a session at American Association of Clinical Endocrinology (AACE) Annual Meeting on 13th May, 2022. Inaugural AAES Guidelines for Adrenalectomy published in JAMA Surgery 2022, includes experts in endocrinology, pathology, radiology & surgery and consists of evidence-based & consensus recommendations for surgery of adrenal disease. Also, routine follow-up of a non-functional adrenal nodule with benign imaging characteristics and non-contrast CT HU.
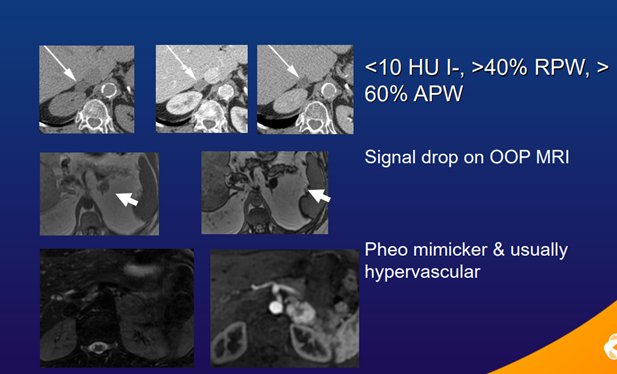
European Society of Endocrinology 2016, recommend establishing if adrenal mass is benign or malignant at the time of detection. If NCCT is consistent with a benign mass (HU≤10) homogeneous & 4cm, no further imaging is required.
If mass is indeterminate on NCCT & negative hormonal work-up, 3 options acknowledging patient’s clinical context: immediate additional imaging with another modality, interval imaging in 6–12months (non-contrast CT or MRI), or surgery without further delay. Imaging can help characterize many adrenal lesions including most incidentalomas. CT and MRI are the main imaging modalities.
Medical societies have some different strategy guidelines particularly regarding follow-up imaging for adrenal mass. Greater harmonization between Imaging and Endocrine Societies could lead to more consistent and optimized clinical practice.
Predictors of Type 2 Diabetes (T2D) Remission using Digital Twin Technology Based on Artificial Intelligence (AI) and Internet of Things (IoT) Technologies
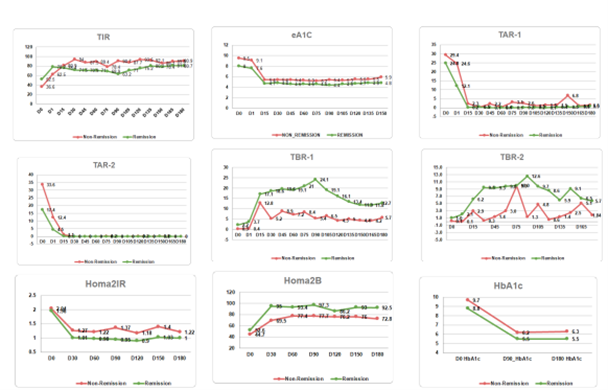
Joshi S, presented a session at American Association of Clinical Endocrinology (AACE) Annual Meeting 2022 on 13th May 2022. The objective of this study is to find pre-intervention anthropometric and biochemical characteristics that predict diabetes remission after using AI-based IoT technology, as well as post-intervention parameters that improve in remission patients.
TPT is a mobile app and intervention based on the proprietary Whole-Body Digital Twin (WBDT) enabled precision treatment. TPT was completed by 167 T2D patients over the course of 180 days. Remission was defined as an A1C result of less than 6.5 % for at least 90 days without the usage of diabetes medication. Using this criterion, 141 of the 167 patients (84.4%) experienced diabetic remission (group R).
The results of the study suggest that patients in the R group had a shorter duration of diabetes at baseline than those in the non-remission Group (NR group). For Time Below Range, the difference in change in R was better than NR after 180 days. Body weight, LDL-C, HDL-C, TGs, and sdLDL levels were all comparable at the start and after the intervention. In NR, the end mean A1C was 6.3 %, compared to 5.5% in R.
The study concludes that the diabetes remission by TPT intervention is reliably predicted and have a shorter duration of diabetes, lower A1C, eA1C, GMI, and TAR2 at baseline. TPT is a promising diabetes remission lifestyle strategy.

