CETP Inhibition: Past Failures, Future Prospects
Dal-GenE: A Pharmaceutical Trial to Reduce Atherosclerotic Events
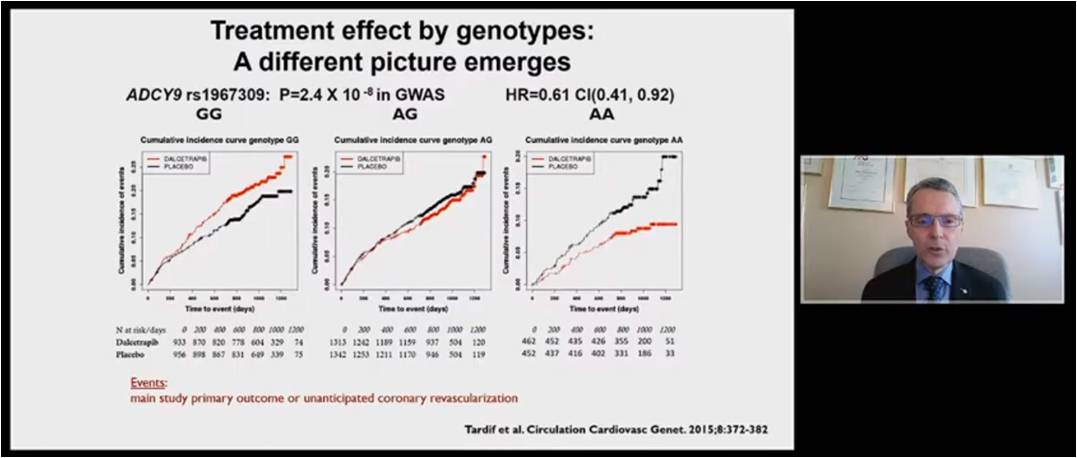
Tardif JC, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. The main Dal-OUTCOMES trial results showed that Dalcetrapib showed no significant difference on the primary end point i.e. composite of death from coronary heart disease, a major nonfatal coronary event, or ischemic stroke as compared to placebo (p=0.52). Discovery GWAS in the dal-OUTCOMES trial showed that a single region with genome-wide significance was correlated with cardiovascular events in the Dalcetrapib group using Cox proportional hazards modeling, identifying SNPs in the ADCY9 (Adenylate Cyclase Type 9) gene on chromosome 16 from 5 ,543,264 common genetic variants. The treatment effect by genotype ADCY9 rs1967309 emerged very different picture in the outcomes in AG patients had neutral impact with Dalcetrapib and placebo whereas AA patients showed significant reduction in the incidence of events with Dalcetrapib (RRR 39%) but GG patients cause harm due to Dalcetrapib as compared to placebo. Dal-Plaque 2 study was independent from Dal-outcomes trial in which 1000 patients were randomized to receive Dalcetrapib showed that the homozygous minor allele patients had regression of atherosclerosis, heterozygous patients had neutral effect and homozygous patients showed major progression of atherosclerosis. Patients with AA and AG genotype showed significant increase in cholesterol efflux than placebo after 12 months whereas patients with GG genotype didn’t show any difference. As per 2018 Circulation paper, ADCY9 genotype inactivation protects from atherosclerosis in mice infected with AAV8-Pcsk9 given a high cholesterol diet. But, ADCY9 inactivation-induced atheroprotection is lost in mice expressing CETP. The recent 2021 study showed the evolutionary interaction between ADCY9 and CETP genotypes on CETP mRNA expression in GEUVADIS group. Meta-analysis supports interaction of CETPi and ADCY9 gene.
A large prospectively conducted clinical trial is required assessing the effects of Dalcetrapib in patients with AA genotype that trial has been existed and conducted from past 4 years called Dal-GenE screened more than 40,000 patients with recent ACS, but failed in GG and AG patients and left with 6147 patients with AA randomized to Dalcetrapib and placebo. The results of the trial will be available in 2022.
A Unified Theory of Cardiovascular and Related Chronic Diseases
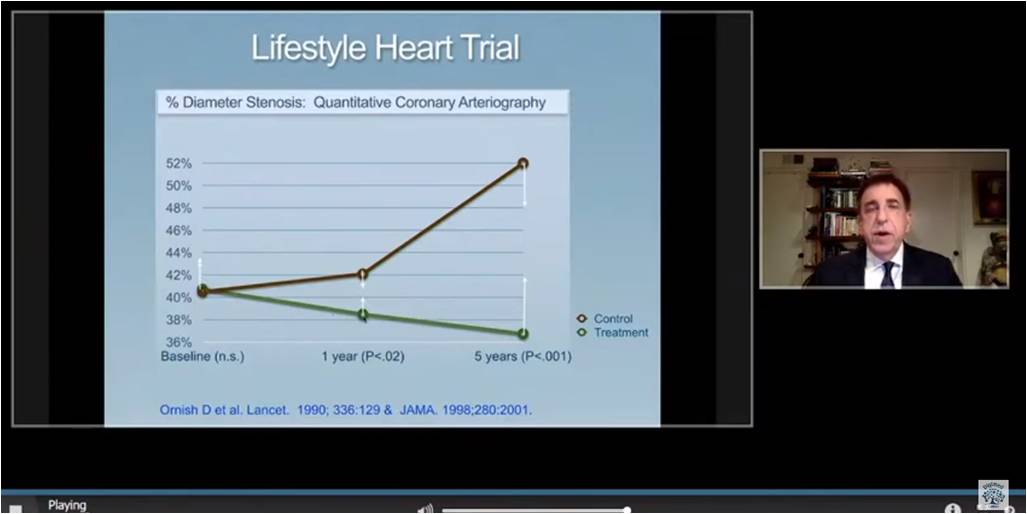
Ornish D, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. A whole food plant-based diet is naturally low in fat and refined carbohydrates. The stress management techniques, moderate exercise and psychological support are important lifestyle medicines. Eat well, move more, stress less and love more are the simple lifestyle changes can be powerful to lead healthy life at low cost. Ornish diet is the best heart-healthy diet from 2011. The same diet and lifestyle can be beneficial for the improvement in health. The reason is that these same lifestyle changes can help prevent and reverse the progression of different chronic diseases is that they are different manifestations of same underlying biological mechanisms. Lifestyle Heart study showed that treatment group had reduction in average percent stenosis from 40.7% at baseline to 38.5% at 1 year to 37.3% at 5 years (p<0.01).
Intensive Cardiac Rehabilitation (ICR) program was conducted for reversing heart disease on 10,180 patients. After nine weeks of ICR program, beneficial results were seen in all biomarkers i.e. reduction in lipid parameters, BP, depression, weight. After 12 weeks, this lifestyle program downregulated genes involved in causing chronic inflammation, oxidative stress, angiogenesis, and atherosclerosis and cholesterol metabolism. Gene expression in 501 genes was beneficially affected in only 3 months. RAS oncogenes that promote prostate cancer and breast cancer were downregulated. The Lancet study showed that positive lifestyle changes may reverse ageing on a cellular level.
The same lifestyles changes affect and may even reverse the progression of a wide range of the most common and costly chronic diseases because they share common biological pathways. These biological pathways are directly influenced by what we eat, how we respond to stress, how much exercise we get and how much love and social support we enjoy. This simplifies the lifestyle recommendations to the patients and to the general public.
Edoxaban Versus Dual Antiplatelet Therapy for Valve Thrombosis and Cerebral Thromboembolism after Transcatheter Aortic-valve Replacement: A Randomized ADAPT-TAVR Trial
Park DW, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. The primary objective of the study was to investigate the effect of Edoxaban compared to DAPT for the prevention of leaflet thrombosis and accompanying potential risks of cerebral thromboembolization and neurological or neurocognitive dysfunction in patients without an OAC indication after TAVR. The secondary objective was to determine the casual association of subclinical leaflet thrombosis with cerebral thromboembolism and neurological/neurocognitive function. In , 220 patients without no indication of OAC after successful TAVR were equally randomized to NOAC (Edoxaban) and DAPT (ASA plus Clopidogrel). The primary endpoint was incidence of leaflet thrombosis on 4D, volume-rendered CT at 6 months. The Edoxaban group did not showed statistical difference in primary end point as compared to DAPT. Lesset no. of patients in the Edoxaban group showed reduced leaflet motion Grade ≥3 than DAPT (p=0.15). Edoxaban group showed increase in presence of new cerebral lesions whereas reduction in median number of total new lesions as compared to DAPT. No difference was seen between both groups in neurological and neurocognitive end points and in safety and efficacy outcomes.
The overall incidence of leaflet thrombosis on CT scans was less frequent with Edoxaban therapy than with the DAPT therapy, although did not reach statistical significance. The incidence of new cerebral thromboembolism on brain MRI and new development of neurological or neurocognitive dysfunction were not different between two groups. There was no association between subclinical leaflet thrombosis and temporally related changes of new cerebral thromboembolic lesions and neurological end points.
Five-year Incidence, Timing And Predictors Of Structural Valve Deterioration of Transcatheter And Surgical Aortic Bioprostheses: Insights From The Corevalve US Pivotal And SURTAVI Trials
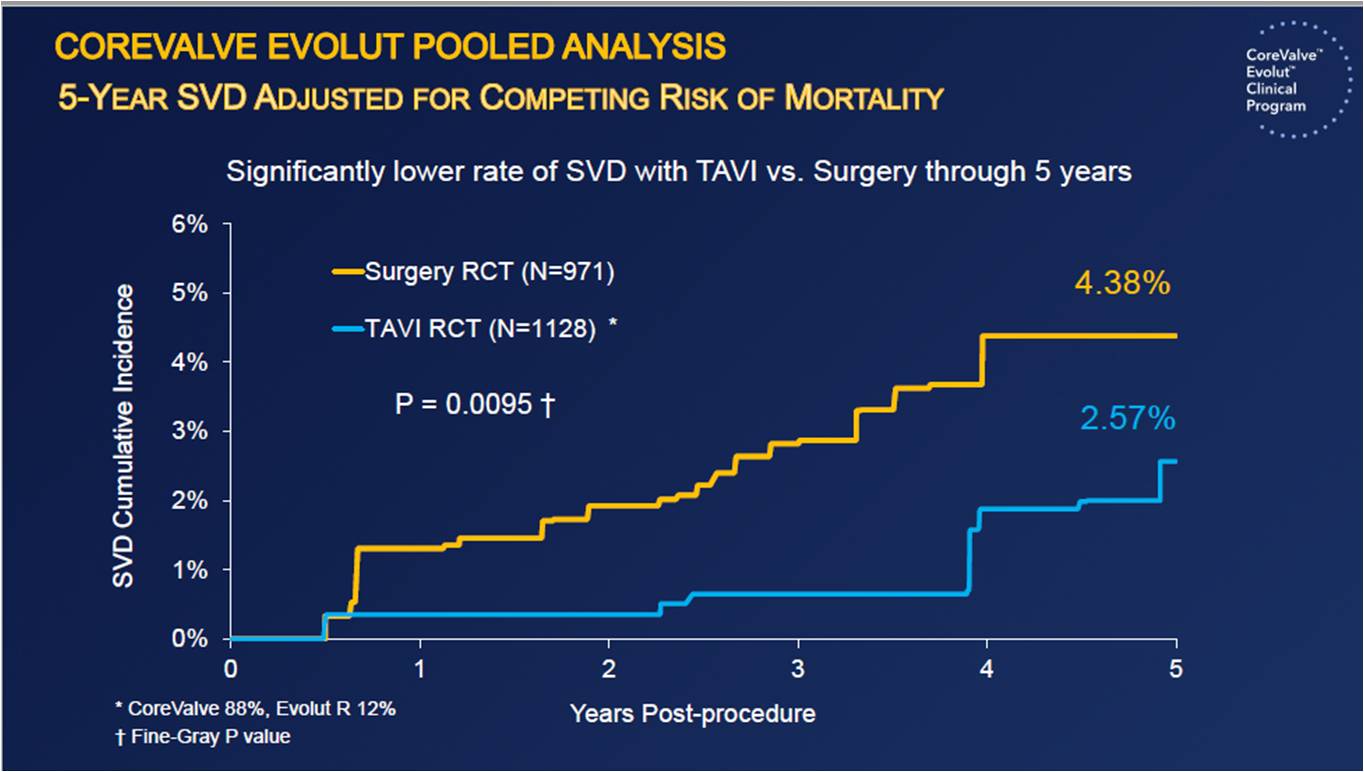
Transcatheter aortic valve implantation (TAVI) is an established treatment for severe aortic stenosis (AS) in patients of all risk levels. Reardon M, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. The objective of the study was to evaluate the 5-year incidence, outcomes and predictors of hemodynamic structural valve deterioration (SVD) in patients undergoing supraannular, self-expanding TAVI and surgery from the Corevalve US pivotal and SURTAVI trials. TAVI showed significantly lower rate of SVD as compared to surgery in 5 years (2.57% vs 4.38%) (p=0.0095). TAVI showed significantly lower rate of SVD in smaller (≤23 mm) annual diameters as compared to surgery in 5 years (1.39% vs 5.86%) (p=0.049). TAVI showed significantly lower rate of SVD in larger (>23 mm) annual diameters as compared to surgery in 5 years (2.48% vs 3.96%) (p=0.067). Corevalve Evolut pooled analysis showed that patients with higher risk of SVD had worsened clinical outcomes i.e mortality and hospitalization.
In patients with severe AS at intermediate or high surgical risk, the 5-year rate of SVD was 4.38% in patients undergoing surgery and 2.57% in patients undergoing TAVI. The difference in SVD was more profound in patients with smaller annuli (5.86% vs 1.39%), but a trend was also found in patient with larger annuli (3.96% vs 2.48%). The Doppler-derived SVD imparted a near 2-fold risk for all-cause mortality and hospitalization for AV disease or worsening HF at 5 years.
A Randomized Trial to Confirm the Safety and Effectiveness of Chocolate Touch Paclitaxel Coated PTA Balloon Catheter in Above the Knee Lesions
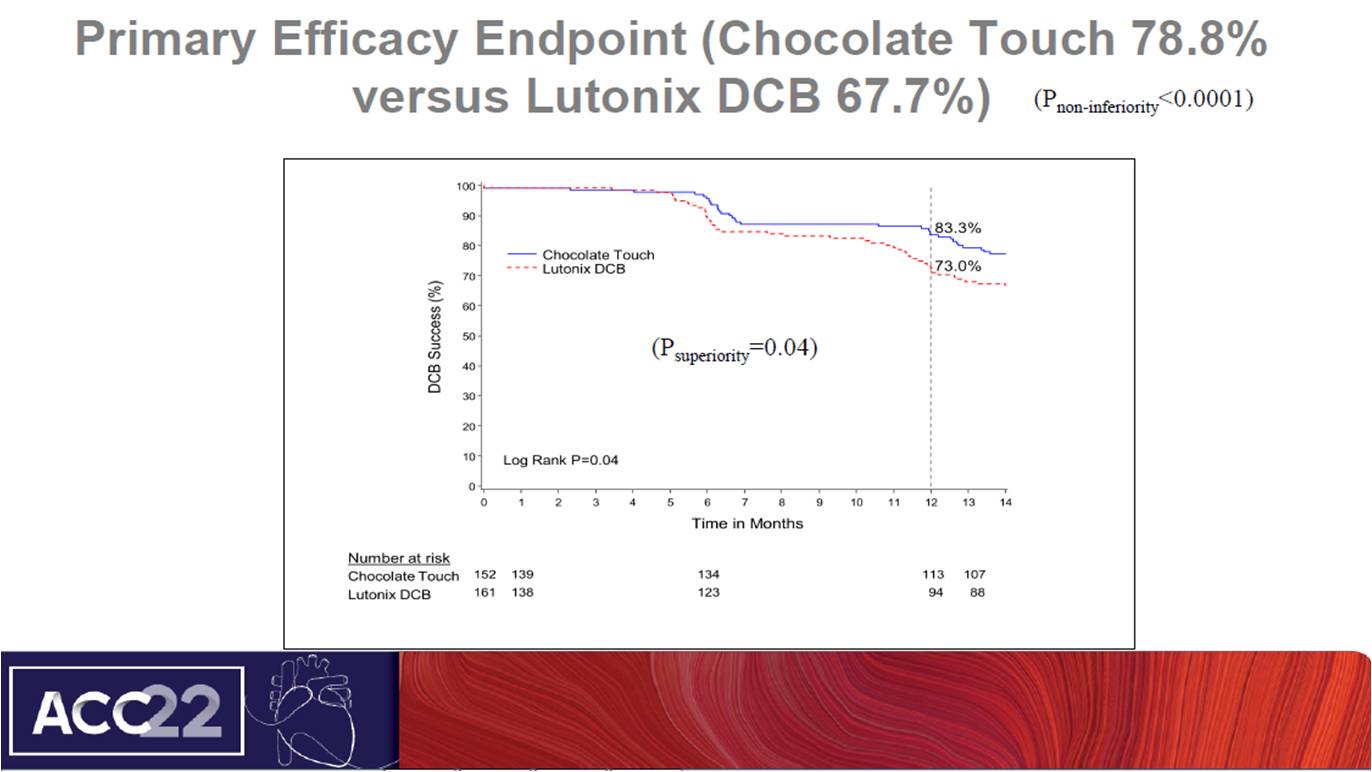
Shishehbor M, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. Chocolate Touch DCB is nitinol constrained balloon designed to reduce vessel trauma and dissections by creating pillow. The distal assembly is coated with paclitaxel to inhibit neointimal formation. The objective of the study was to compare the efficacy and safety of the chocolate touch DCB to the commercially approved lutonix DCB in an international randomized clinical trial. Chocolate Touch, an open-label, randomized, non-inferiority trial in which patients with symptomatic SFA or popliteal arteries were randomized to chocolate touch group (n=152) and Lutonix (n=161). The primary effectiveness endpoint was true DCB success at 12 months and safety endpoint was freedom from major adverse events (MAE) at 12 months. At follow up of 12 months, 142 patients of chocolate touch group and 149 patients of Lutonix group completed the visits. Chocolate touch group showed significant improvement in primary efficacy endpoint as compared to lutonix (78.8% vs 67.7%) (pnon-inferiority<0.0001). Chocolate touch group showed consistent true DCB success at 12 months. At primary safety endpoint, both groups showed no significance difference and lead to non-inferiority. Chocolate touch group showed similar mortality as treated population.
The Chocolate Touch study met its primary effectiveness of True DCB success at 12 months with superior efficacy than Lutonix group. Chocolate Touch also met its non-inferiority endpoint for safety. No difference in mortality is seen, although the trial was not adequately powered for a mortality endpoint.
Very Mild Perioperative Hypothermia versus Aggressive Warming and Myocardial Injury after Non-Cardiac Surgery: The PROTECT Trial
Sessler D, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. It has been reported by small trial that mild hypothermia (≈35.5°C) results in increased morbid myocardial outcomes. Other trials have also reported that moderate perioperative hypothermia (≈34.5°C) can promote surgical site infections and increase transfusion requirements. As per the international guidelines, it is recommended to keep surgical patients normothermic and active intraoperative warming. The following study primarily hypothesised that aggressive intraoperative warming reduces the incidence of major cardiovascular complications.
The study was conducted as a multi-centre trial in which patients were randomly assigned to routine care (intraoperative core temperature ≈35.5°C) or aggressive warming (≈37°C). The primary outcome assessed was a composite of myocardial injury (troponin elevation, judged due to ischemia), non-fatal cardiac arrest, and all-cause mortality within 30 days of surgery.
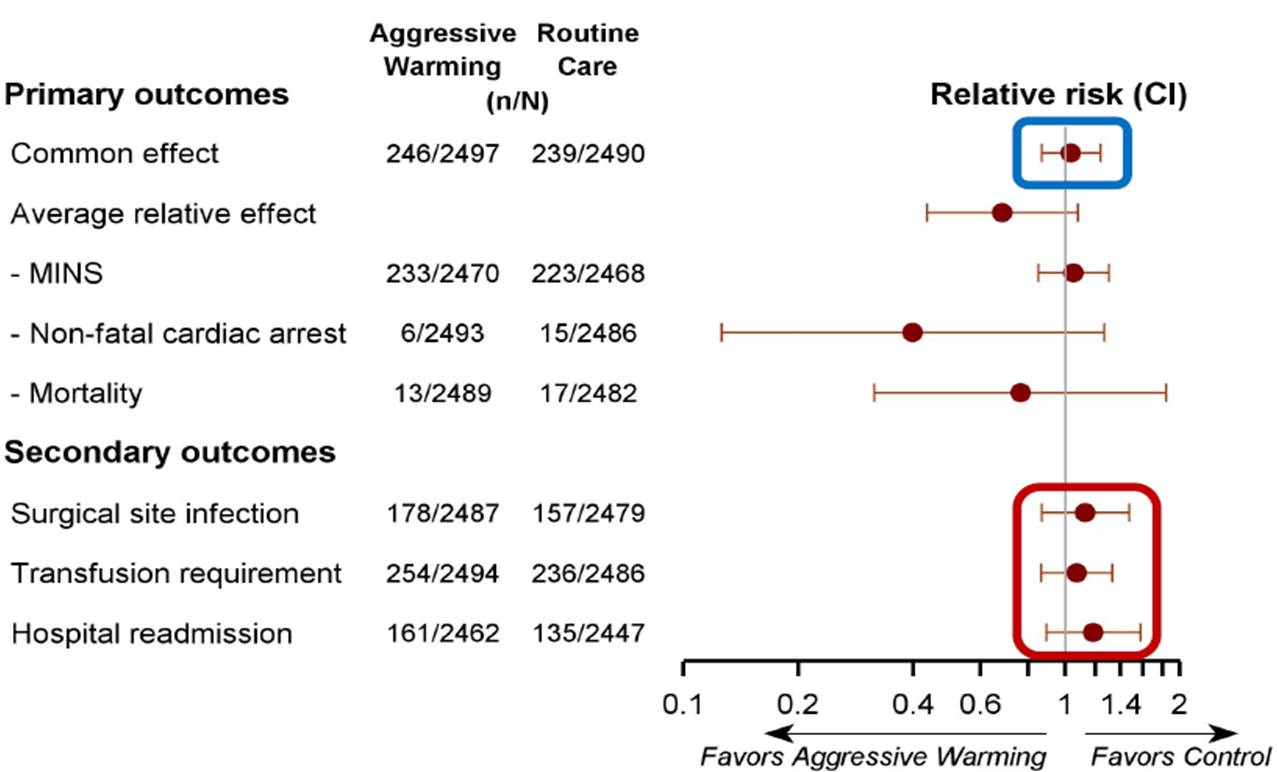
In this multicentre trial, a total of 5,050 patients were randomized and the mean age was 67 years; a third were female. Final intraoperative temperatures were 35.6±0.3 (SD) and 37.1±0.3°C. The number of cardiovascular complications was found to be similar in patients randomized to 35.5°C and to 37°C: 218/2507 v. 226/2505 patients: RR 1.04 [95% CI: 0.87, 1.25]. At each temperature, serious wound infections [RR 1.13 (95% CI: 0.87, 1.47)], transfusions [RR 1.07 (0.86, 1.32)], duration of hospitalization [HR 0.98 (0.91, 1.05)] and hospital re-admissions [RR 1.19 (0.89, 1.57)] were also similar.
The study concludes that size of PROTECT trial is 10 times more than ten times the size of previous thermoregulatory trials, most of which were conducted two decades ago. Over a 1.5°C range from very mild hypothermia to full normothermia, no outcome varied and keeping the temperature at ≥35.5°C in surgical patients appears sufficient.
Long Term Effect of Renal Denervation on Blood Pressure Reduction in Patients on Antihypertension Medications: 3-year Efficacy Outcomes from the SPYRAL HTN-ON MED Trial

Mahfoud F, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. In SPYRAL HTN-ON Pilot, it was found that renal denervation (RDN) significantly lowered blood pressure (BP) at 6 months in patients with uncontrolled hypertension (HTN) on antihypertension (AH) drugs.
In this trial, patients were randomized in 1:1 to undergo RDN or a sham procedure. The eligible patients had an office systolic BP of 150 to <180 mmHg, diastolic BP >90 mmHg, 24h ambulatory systolic BP (ASBP) of 140 to <170 mmHg, and were administered with 1-3 AH drugs. The study enrolled a total of 80 participants with uncontrolled HTN (n=38 RDN, n=42 sham).
The results of the study show that at the end of 3 years, RDN patients showed greater ABSP reduction compared to sham control patients (-18.7±12.4 mmHg vs. -8.6±14.6 mmHg, p=0.004, respectively and office BP systolic reductions showed a similar trend at 3 years (-20.9±15.3 mmHg vs. -12.5±21.6 mmHg, p=0.073).
Radio frequency RDN patients compared with sham control patients showed significant reductions in ambulatory systolic BP independent of concomitant antihypertensive medications out to 3 years despite similar AH medication burden. Given the long term safety and efficacy, RDN provides an adjuctive treatment modality in the management of hypertension.
Differential Impact of Residual Inflammatory Risk and Residual Cholesterol Risk Among Atherosclerosis Patients with And Without Chronic Kidney Disease: Secondary Analysis of The CANTOS Trial
Ridker P, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. Atherosclerotic disease is jointly contributed by hyperlipidaemia and inflammation and relative contributions of these processes differs in patient groups, with and without patient groups. In CANTOS, 9,151 stable statin treated post-myocardial infarction patients were included to compare the relative contributions of residual cholesterol risk and residual inflammatory risk as determinants of recurrent major adverse cardiovascular events (MACE).
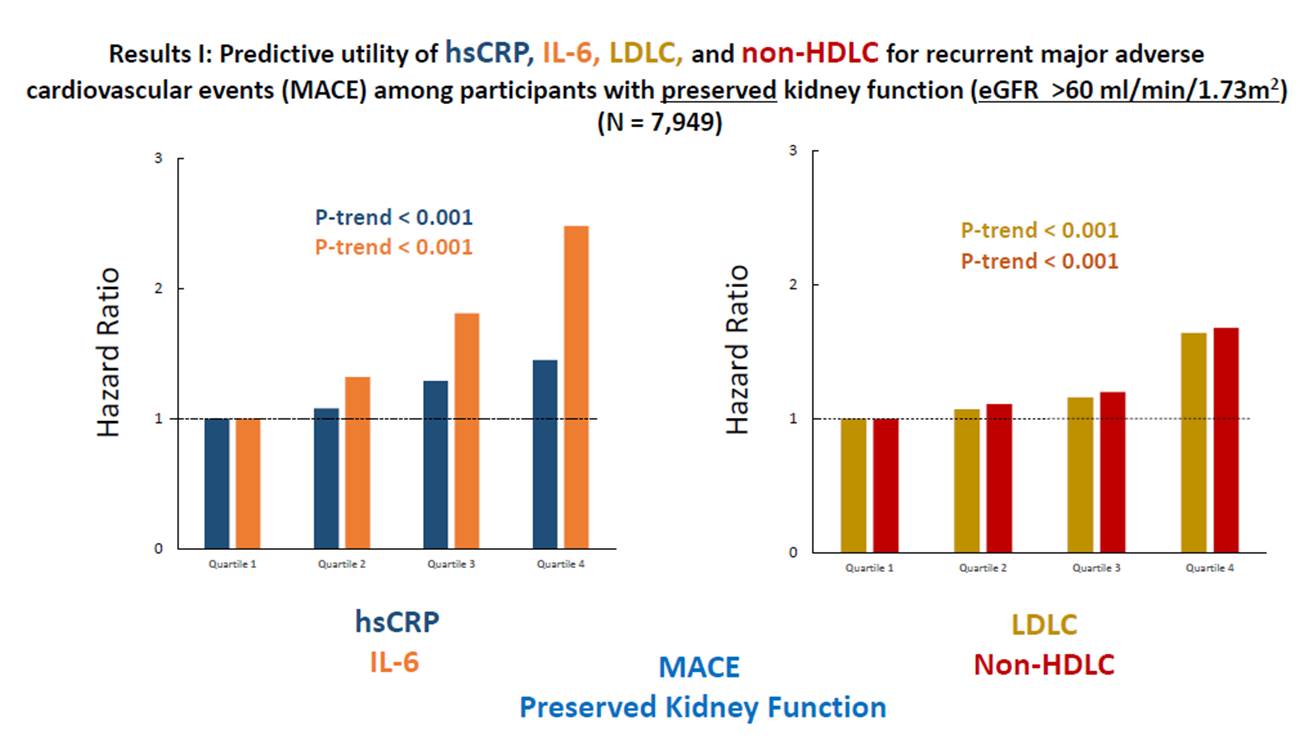
The patients in CANTOS were stratified by baseline estimated glomerular filtration rate (eGFR) above or below 60 mL/min/1.73m2 and follow up was done for 5 years. Among CANTOS participants with eGFR >60 mL/min/1.73m2, there was a positive association between plasma hsCRP, IL-6, LDLC, and non-HDLC and risks of recurrent MACE (all P-values <0.0001). Among participants with eGFR <60 mL/min/1.73m2, increasing quartiles of hsCRP [P=0.006] and IL-6 [P=0.018]) significantly predicts recurrent MACE.
After adjustment for a wide range of potential confounding factors, the predictive utility of inflammation as compared to hyperlipidemia among patients with eGFR <60 mL/min/1.73m2 remained significant and was particularly striking for cardiovascular and all-cause mortality.
Among atherosclerotic patients with impaired kidney function, residual inflammatory risk may hold a greater importance than residual cholesterol risk. The data available from CANTOS can help in the development of novel agents that target inflammatory processes in this high-risk group of patients.
Effects of Empagliflozin on Symptoms, Physical Limitations and Quality of Life in Patients Hospitalized for Acute Heart Failure- Results from the EMPULSE Trial
Kosiborod M, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. Heart failure patients are associated with high burden of symptoms, physical limitations and poor quality of life. SGLT2 inhibitors causes reduction in symptoms in chronic HF, but their effects in acute heart failure are not well characterized.
In this trial, patients with AHF (de novo or decompensated chronic HF, regardless of ejection fraction or diabetes) were randomized to empagliflozin (EMPA) 10 mg daily or placebo (PBO) for 90 days. Assessment of Kansas City Cardiomyopathy Questionnaire (KCCQ) was done at baseline, 15, 30 and 90 days. Analysis of Change (baseline to Day 90) in KCCQ Total Symptom (TSS), Physical Limitations (PLS), quality of life (QoL), clinical summary (CSS) and overall summary (OSS) scores was done.
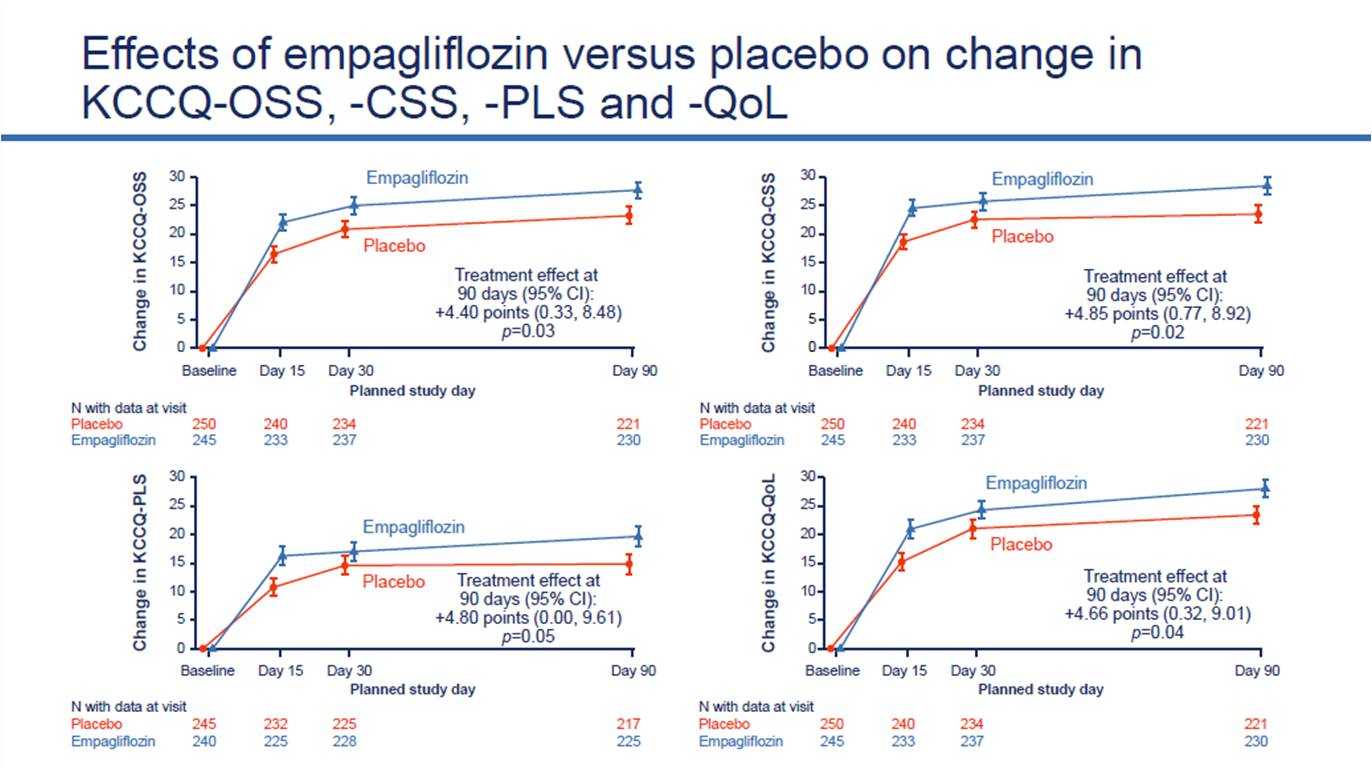
A total of 530 patients were randomized with 265 patients in each arm. EMPA-treated pts had a greater improvement in KCCQ TSS, PLS, QoL, CSS and OSS at Day 90 [effect sizes (95% CI): 4.5 (0.3, 8.6), p = 0.035; 4.80 (0.0, 9.6), p = 0.050; 4.7 (0.3, 9.0), p = 0.036; 4.9 (0.8, 8.9), p = 0.02; and 4.4 (0.3, 8.5), p = 0.03, respectively]. Benefits were evident at 15 days and were maintained throughout 90 days.
The study concludes that initiation of EMPA in patients hospitalized for AHF showed improvement in symptoms, physical limitations and quality of life with benefits seen at 15 and maintained through 90 days.
Comparison of Fractional Flow Reserve-guided And Intravascular Ultrasoundguided Percutaneous Coronary Intervention in Intermediate Coronary Artery Stenosis: The FLAVOUR Randomized Clinical Trial
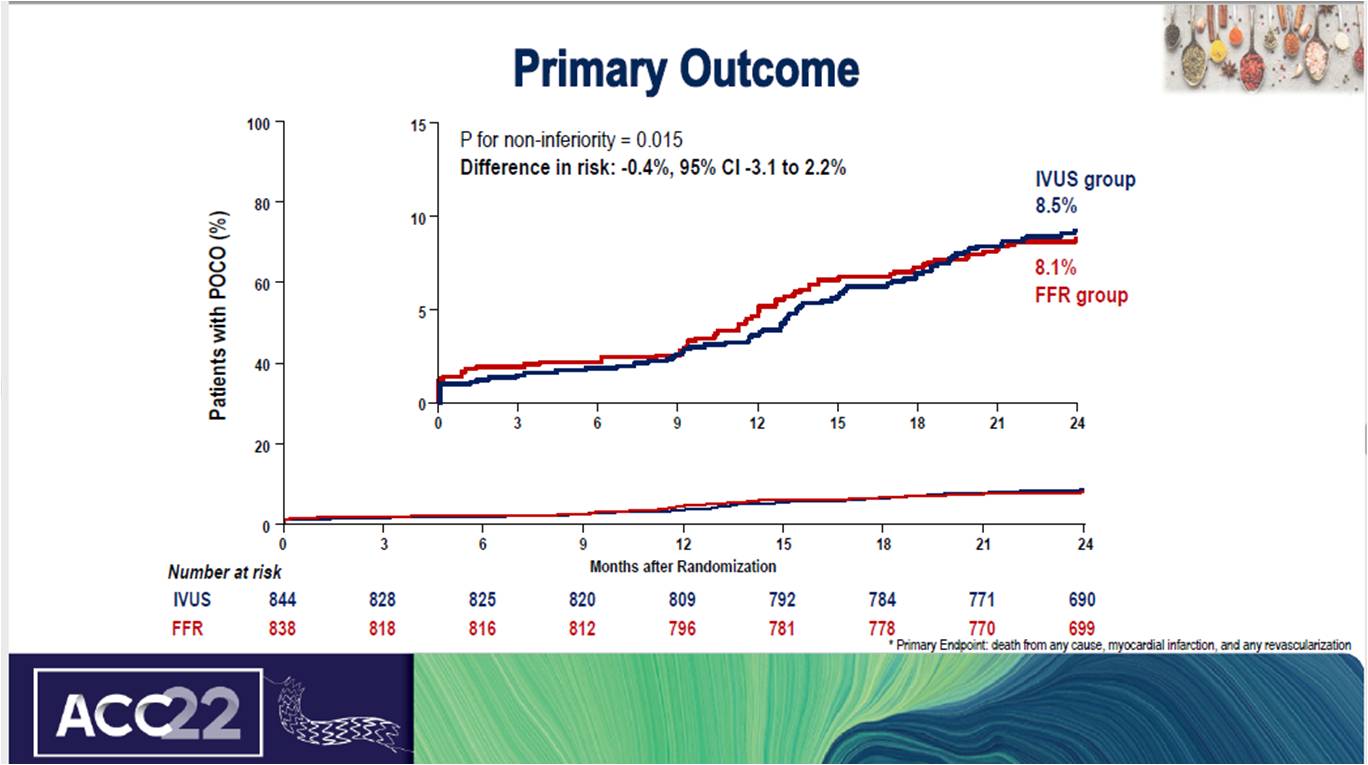
Koo B, presented a study in a session at American College of Cardiology (ACC) on 4th April, 2022. The current standard to define the presence of ischemia in patients with coronary artery disease is fractional flow reserve (FFR) but clinical events still occur after FFR-guided deferral of revascularization. To reduce the risk of adverse events, IVUS-guided percutaneous coronary intervention is most commonly used in comparison with angiography-guided PCI. However, there has not been a solid evaluation about the impact of FFR-guided vs. IVUS-guided PCI on clinical outcomes.
Patients with intermediate stenosis were randomized to either FFR-guided or IVUS-guided PCI in a 1:1 ratio in this international multi-centre study. In the FFR group, PCI was performed according to the predefined criteria for revascularization; FFR ≤ 0.80. In the IVUS group, the predefined criteria were minimal lumen area (MLA) ≤3 mm2 or 3 mm2 < MLA ≤4 mm2 and plaque burden >70%. Primary endpoints assessed were composite of all-cause death, myocardial infarction, and any revascularization at 24 months.
A total of 1682 patients were successfully randomized with the mean age of 65 years. In the FFR group, the rate of target lesion PCI was significantly lower than in the IVUS group (32.9% vs. 57.6%, p<0.001). At the end of 2 years, the primary composite endpoint rate was 8.1% in the FFR group and 8.6% in the IVUS group (hazard ratio, 0.94; 95% CI, 0.67 to 1.31).
The study concludes that FFR group is non-inferior to IVUS group with regards to adverse clinical outcomes during the 2-year follow-up period. Owing to different criteria for revascularization, the FFR group received lesser stents than the IVUS group.

