
On December 30, 2025, Vanda Pharmaceuticals announced that the U.S. Food and Drug Administration (FDA) approved NEREUS™ (tradipitant), an oral neurokinin-1 (NK-1) receptor antagonist, for the prevention of vomiting induced by motion in adults. This approval represents a historic milestone, as it introduces the first new pharmacologic treatment for motion sickness in over 40 years, advancing beyond traditional antihistamines and antimuscarinics like scopolamine patches, which often carry limitations such as drowsiness or limited efficacy.

Injectable GLP-1 receptor agonists like semaglutide (Wegovy® 2.4 mg weekly) have transformed obesity treatment, demonstrating substantial weight loss and cardiovascular risk reduction. However, patient preference for oral administration and potential barriers to injectable therapies highlight the need for non-injectable options. Novo Nordisk developed a once-daily oral formulation of semaglutide at 25 mg for weight management.
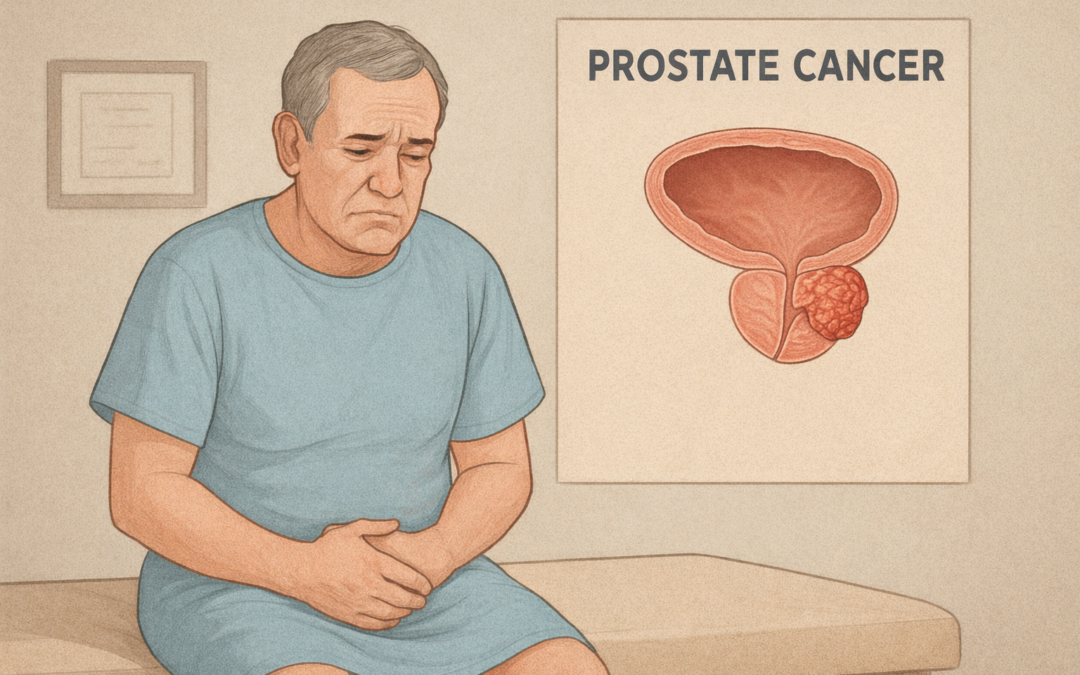
On December 12, 2025, the U.S. Food and Drug Administration (FDA) granted approval for AKEEGA® (niraparib and abiraterone acetate dual-action tablet) in combination with prednisone for the treatment of adult patients with deleterious or suspected deleterious BRCA2-mutated metastatic castration-sensitive prostate cancer (mCSPC). This supplemental New Drug Application (sNDA) approval marks AKEEGA as the first and only precision medicine combination of a PARP inhibitor and androgen receptor pathway inhibitor specifically indicated for BRCA2-mutated mCSPC, expanding its prior 2023 approval for BRCA-mutated metastatic castration-resistant prostate cancer (mCRPC).
Hypertension remains a global health crisis, affecting over 1.4 billion people and serving as a leading risk factor for cardiovascular and renal diseases. Despite advances in antihypertensive therapies, approximately 50% of treated patients in the US fail to achieve blood pressure control with multiple agents, highlighting a critical unmet need in hard-to-control (uncontrolled or treatment-resistant) hypertension. Aldosterone, a hormone that promotes sodium and water retention, plays a pivotal role in this resistance, yet targeted interventions have been limited for over two decades.
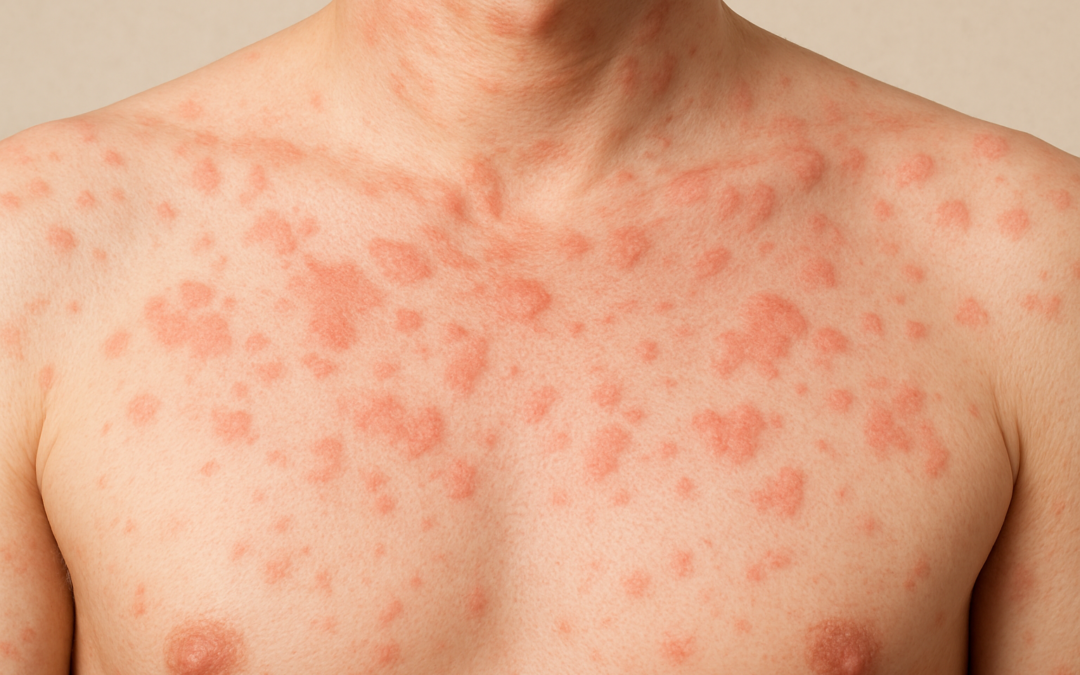
Chronic spontaneous urticaria (CSU) affects approximately 270,000 adults and adolescents aged 12 and older in the European Union, manifesting as unpredictable outbreaks of debilitating hives and intense itch that disrupt daily life. For many, standard first-line treatments like H1 antihistamines provide insufficient relief, leaving patients in cycles of discomfort and frustration. In a landmark development, Sanofi and Regeneron Pharmaceuticals have secured European Commission approval for Dupixent (dupilumab), marking the first targeted biologic therapy for moderate-to-severe CSU in over a decade.

Muscle-invasive bladder cancer (MIBC) represents approximately 30% of the ~614,000 annual global bladder cancer cases, ranking it as the ninth most common malignancy worldwide. While neoadjuvant cisplatin-based chemotherapy followed by cystectomy remains the standard for eligible patients, up to half of those with MIBC are ineligible for cisplatin due to comorbidities, renal impairment, or other factors, leaving them with limited perioperative options and high recurrence rates—nearly 50% even post-surgery. On November 21, 2025, the U.S. Food and Drug Administration (FDA) approved a transformative regimen: PADCEV® (enfortumab vedotin-ejfv), a Nectin-4-directed antibody-drug conjugate (ADC), in combination with Keytruda® (pembrolizumab) or Keytruda QLEX™ (pembrolizumab and berahyaluronidase alfa-pmph), for neoadjuvant treatment followed by adjuvant therapy post-cystectomy in cisplatin-ineligible adults with MIBC.







