
A large population-based screening study published in JAMA examined more than 220,000 children in Germany to detect early, presymptomatic type 1 diabetes using islet autoantibody testing. The study aimed to understand how common early-stage disease is and how it progresses over time in the general pediatric population.
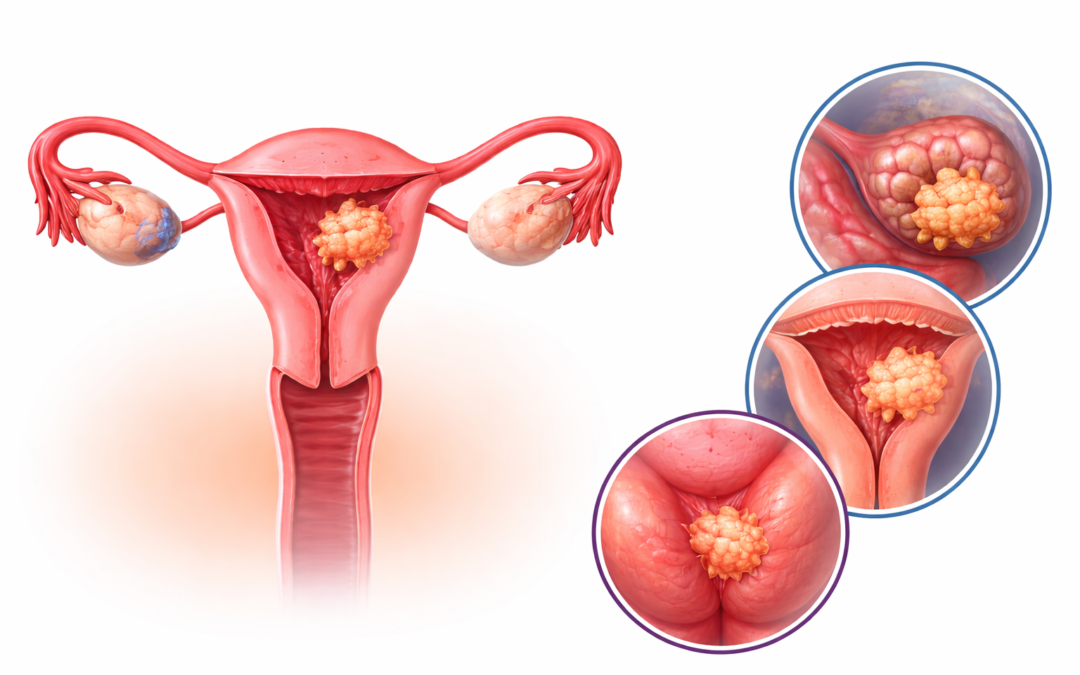
GSK has reported promising clinical data for its investigational B7-H4-targeted antibody–drug conjugate (ADC), mocertatug rezetecan (also known as GSK5733584), in patients with gynaecological cancers, including ovarian and endometrial malignancies. These cancers often present at advanced stages and are associated with poor prognosis, particularly in patients who develop resistance to standard therapies such as platinum-based chemotherapy. The emergence of targeted therapies like ADCs represents a significant advancement in addressing these unmet clinical needs.

The emergence of oral glucagon-like peptide-1 (GLP-1) receptor agonists marks a significant advancement in obesity pharmacotherapy, offering needle-free alternatives to injectable agents. In this context, a new population-adjusted indirect treatment comparison (ITC), to be presented at the Obesity Medicine Association (OMA) 2026 annual meeting, evaluated the relative efficacy and tolerability of oral semaglutide 25 mg (Wegovy® pill) versus orforglipron 36 mg.

The role and optimal duration of beta-blocker therapy following myocardial infarction (MI) have been increasingly questioned in the contemporary era of advanced reperfusion strategies and secondary prevention therapies. In a recent study published in the New England Journal of Medicine, investigators evaluated the safety and clinical outcomes associated with discontinuation of long-term beta-blocker therapy in stable post-MI patients without clear ongoing indications such as heart failure or reduced left ventricular ejection fraction (LVEF).
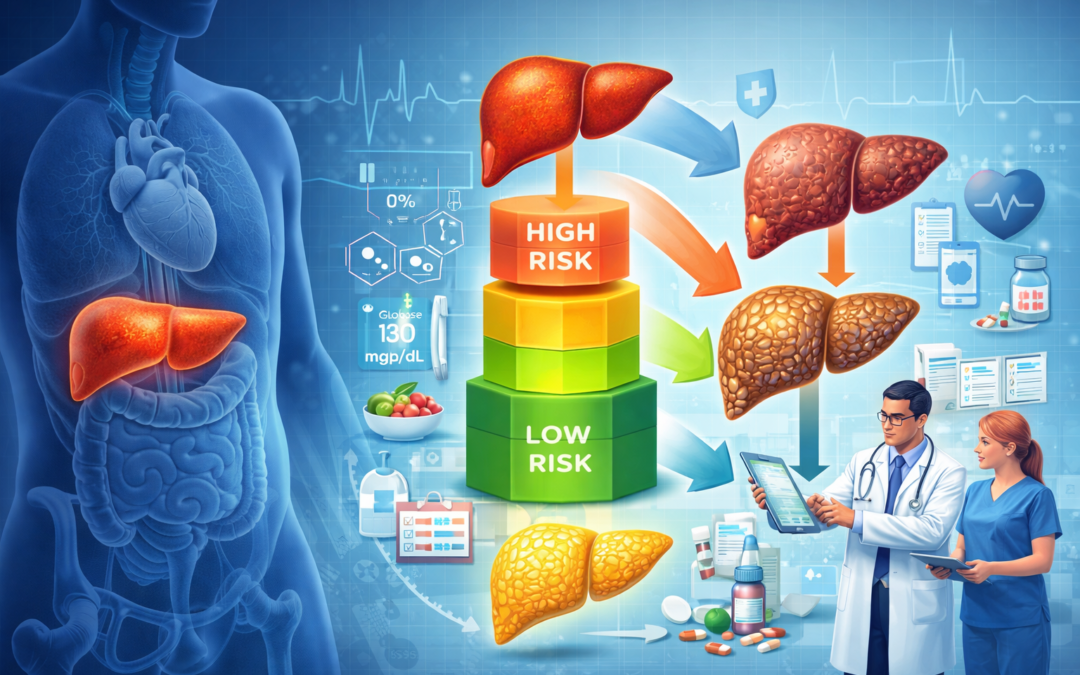
The newly proposed clinical care pathway for metabolic dysfunction-associated steatotic liver disease (MASLD) represents an important step toward standardizing the evaluation and management of one of the most prevalent chronic liver diseases globally. MASLD, closely linked to obesity, type 2 diabetes, and other cardiometabolic risk factors, is frequently managed in primary care and endocrinology settings, necessitating practical, scalable approaches to care.

Novo Nordisk has received US regulatory approval for Wegovy® HD (semaglutide 7.2 mg), representing a significant advancement in pharmacological obesity management. This higher-dose formulation of the GLP-1 receptor agonist semaglutide is designed to address unmet needs in patients who do not achieve adequate weight loss with existing therapies. The approval is supported by robust data from the phase 3 STEP UP clinical trial program, which evaluated the efficacy and safety of once-weekly semaglutide 7.2 mg in adults with obesity.

The 2026 consensus statement from the American Association of Clinical Endocrinology reflects a continued evolution in endocrine care, emphasizing a comprehensive, patient-centered, and multidisciplinary framework for managing cardiometabolic diseases. Building on prior AACE guidance and algorithms, the statement integrates the latest evidence with practical clinical strategies to address conditions such as obesity, diabetes, dyslipidemia, and related metabolic disorders.

The 2026 ACC/AHA/AACVPR/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Dyslipidemia represents a major update in lipid management, integrating contemporary evidence to optimize prevention of atherosclerotic cardiovascular disease (ASCVD). The guideline provides a comprehensive, life-course approach to the evaluation, treatment, and monitoring of dyslipidemias, encompassing both primary and secondary prevention strategies.

Psoriatic arthritis (PsA) is a chronic immune-mediated inflammatory disease characterized by joint inflammation, structural damage, and associated skin manifestations of psoriasis. Despite the availability of several biologic and targeted therapies, many patients continue to experience persistent disease activity or treatment intolerance, highlighting the need for additional therapeutic options. Deucravacitinib (Sotyktu), an oral selective tyrosine kinase 2 (TYK2) inhibitor developed by Bristol Myers Squibb, has now received approval from the U.S. Food and Drug Administration for the treatment of adults with active psoriatic arthritis. This approval represents the first TYK2 inhibitor authorized for this indication and introduces a novel mechanism targeting intracellular cytokine signaling pathways involved in immune-mediated inflammation.

Duchenne muscular dystrophy (DMD) is a severe X-linked neuromuscular disorder characterized by progressive muscle degeneration caused by mutations in the dystrophin gene. Z-rostudirsen (DYNE-251) is an investigational exon-51–skipping therapy designed to restore the production of functional dystrophin by enabling targeted delivery of a phosphorodiamidate morpholino oligomer to muscle tissue. Dyne Therapeutics recently reported new cardiopulmonary and long-term clinical results from the Phase 1/2 DELIVER trial evaluating this therapy in individuals with DMD amenable to exon 51 skipping.











