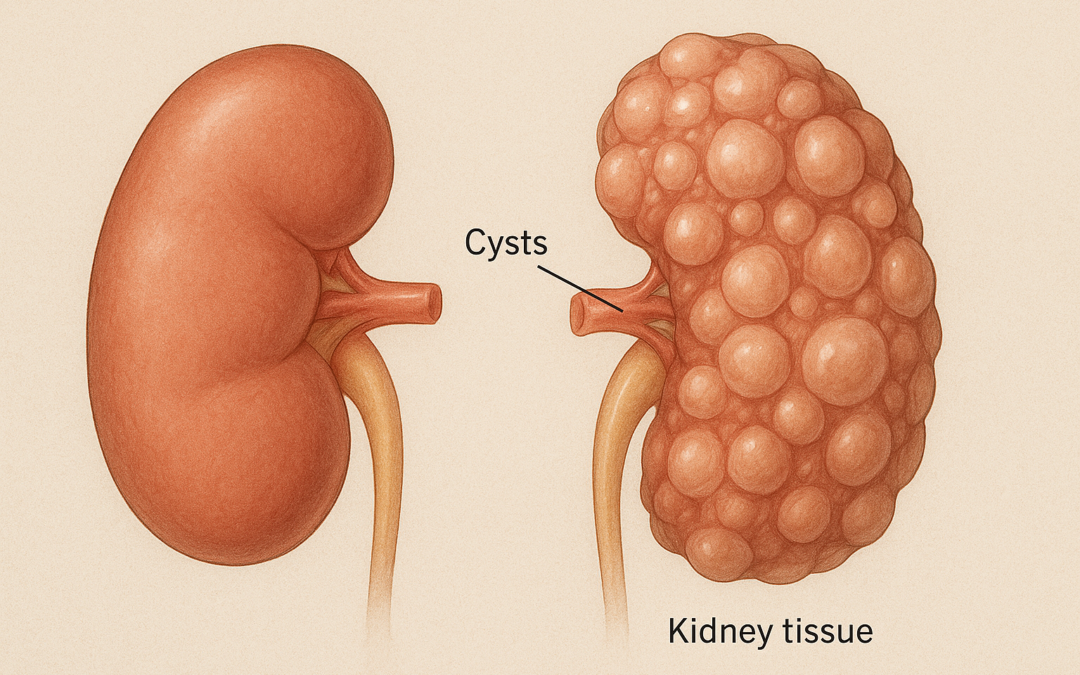
Autosomal dominant polycystic kidney disease (ADPKD) is a leading cause of kidney failure, necessitating novel therapies to slow disease progression. The Implementation of Metformin Therapy to Ease Decline of Kidney Function in Polycystic Kidney Disease (IMPEDE-PKD) trial is a prospective, multi-centre, international, double-blind, randomized, placebo-controlled phase III study evaluating metformin’s efficacy in reducing kidney function decline in ADPKD patients.
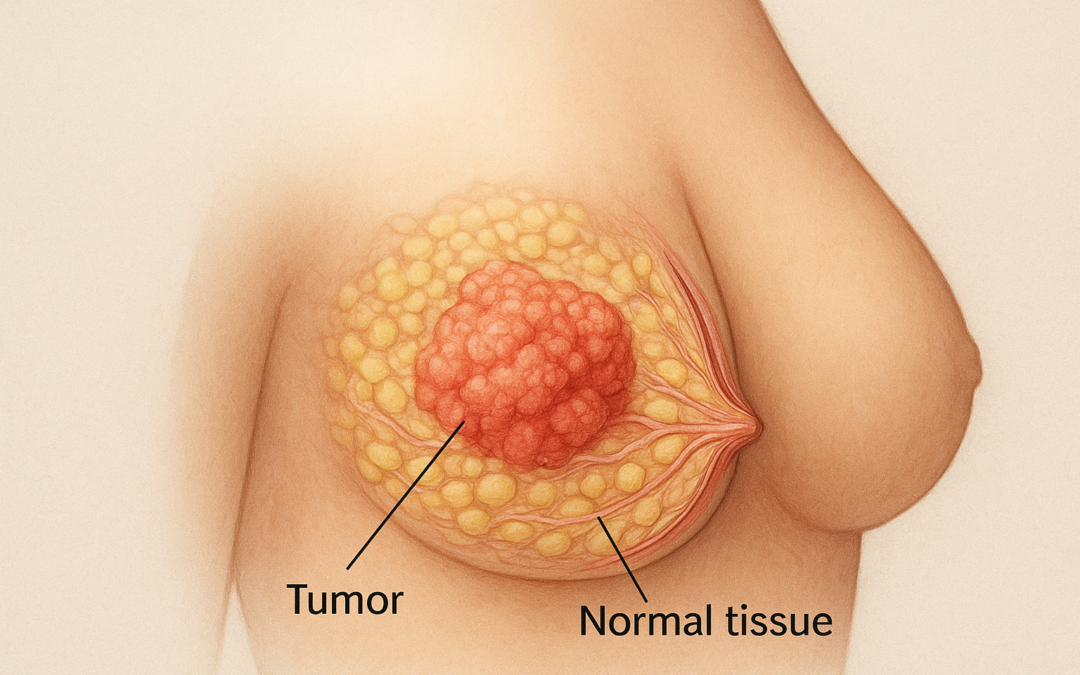
Eli Lilly and Company’s Verzenio® (abemaciclib), a CDK4/6 inhibitor, has demonstrated significant improvement in overall survival (OS) in patients with hormone receptor-positive (HR+), HER2-negative (HER2-), node-positive, high-risk early breast cancer, as reported in the phase 3 monarchE trial. This global, randomized, open-label, multicenter study enrolled 5,637 patients across 600 sites in 38 countries, evaluating two years of Verzenio plus endocrine therapy (ET) against ET alone.

This prospective cohort study investigated the longitudinal impact of semaglutide, a glucagon-like peptide-1 receptor agonist (GLP-1RA), on sarcopenia indicators and biomarkers of neuromuscular junction (NMJ) stability and neuronal health in older men with type 2 diabetes mellitus (T2DM), compared to sitagliptin as a control. Older adults with T2DM are at elevated risk for sarcopenia, characterized by loss of muscle mass, strength, and function, which can exacerbate disability and reduce quality of life.
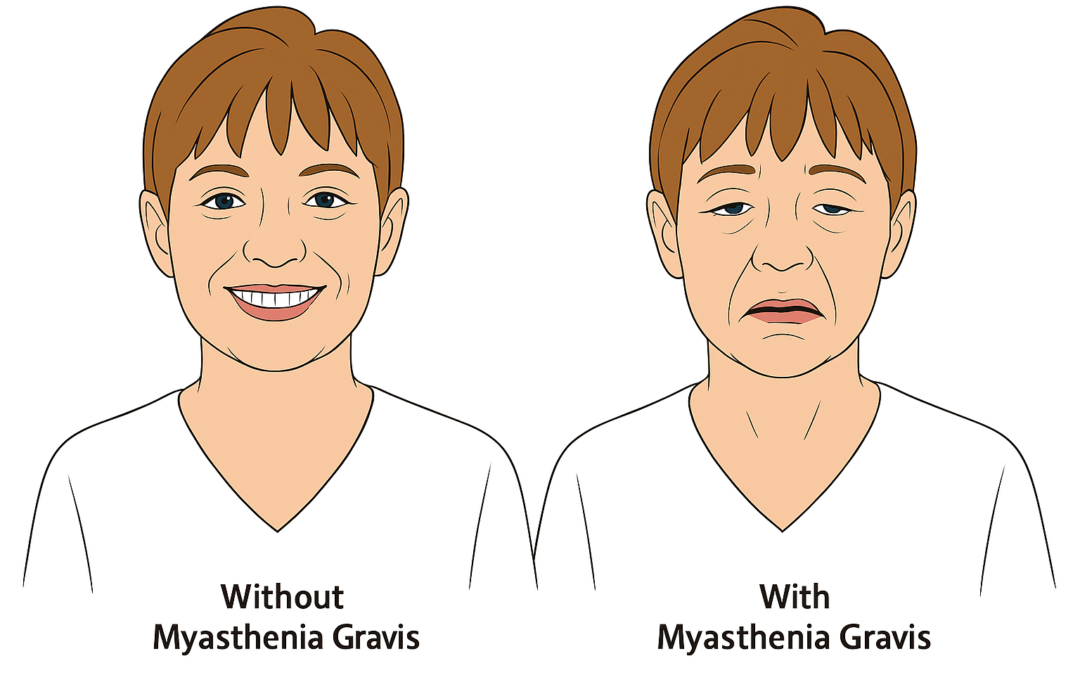
Regeneron Pharmaceuticals reported positive topline results from the Phase 3 NIMBLE trial evaluating investigational cemdisiran, an siRNA targeting complement factor 5 (C5), as monotherapy for adults with generalized myasthenia gravis (gMG). The trial, involving patients with anti-acetylcholine receptor antibodies, randomized participants to receive cemdisiran (600 mg subcutaneously every 12 weeks), a cemdisiran-pozelimab combination (200 mg each every 4 weeks), or placebo.
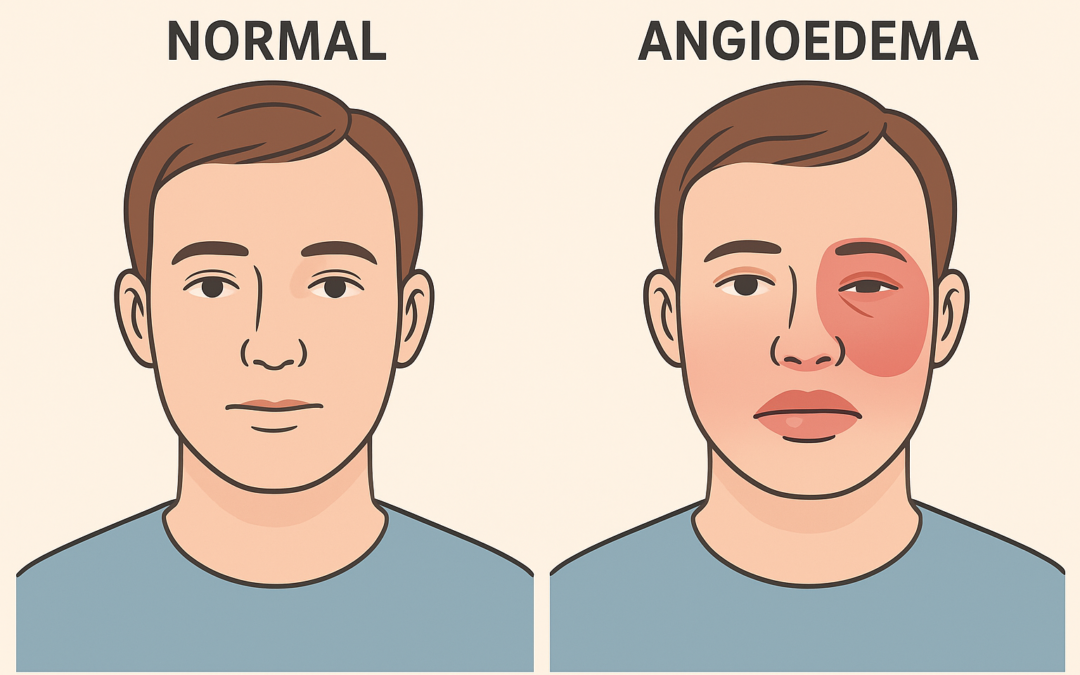
Ionis Pharmaceuticals, Inc. received FDA approval on August 21, 2025, for DAWNZERA™ (donidalorsen), the first RNA-targeted therapy for prophylaxis against hereditary angioedema (HAE) attacks in adults and pediatric patients aged 12 and older. HAE, a rare genetic condition affecting approximately 7,000 Americans, causes recurrent, potentially life-threatening swelling in various body parts. DAWNZERA targets plasma prekallikrein (PKK), a key protein in HAE attack pathways, and is administered via subcutaneous autoinjector every four (Q4W) or eight weeks (Q8W), offering the longest dosing interval among HAE prophylactics. This approval marks Ionis’ second independent product launch within nine months, following TRYNGOLZA® for familial chylomicronemia syndrome.






