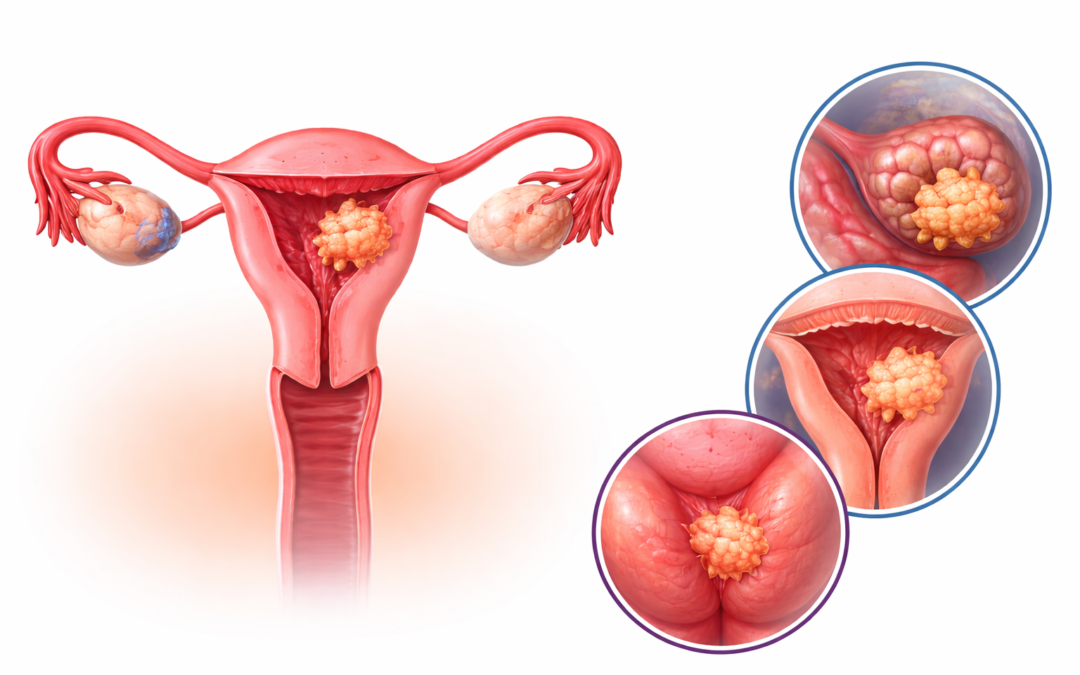
GSK has reported promising clinical data for its investigational B7-H4-targeted antibody–drug conjugate (ADC), mocertatug rezetecan (also known as GSK5733584), in patients with gynaecological cancers, including ovarian and endometrial malignancies. These cancers often present at advanced stages and are associated with poor prognosis, particularly in patients who develop resistance to standard therapies such as platinum-based chemotherapy. The emergence of targeted therapies like ADCs represents a significant advancement in addressing these unmet clinical needs.

The emergence of oral glucagon-like peptide-1 (GLP-1) receptor agonists marks a significant advancement in obesity pharmacotherapy, offering needle-free alternatives to injectable agents. In this context, a new population-adjusted indirect treatment comparison (ITC), to be presented at the Obesity Medicine Association (OMA) 2026 annual meeting, evaluated the relative efficacy and tolerability of oral semaglutide 25 mg (Wegovy® pill) versus orforglipron 36 mg.

The role and optimal duration of beta-blocker therapy following myocardial infarction (MI) have been increasingly questioned in the contemporary era of advanced reperfusion strategies and secondary prevention therapies. In a recent study published in the New England Journal of Medicine, investigators evaluated the safety and clinical outcomes associated with discontinuation of long-term beta-blocker therapy in stable post-MI patients without clear ongoing indications such as heart failure or reduced left ventricular ejection fraction (LVEF).
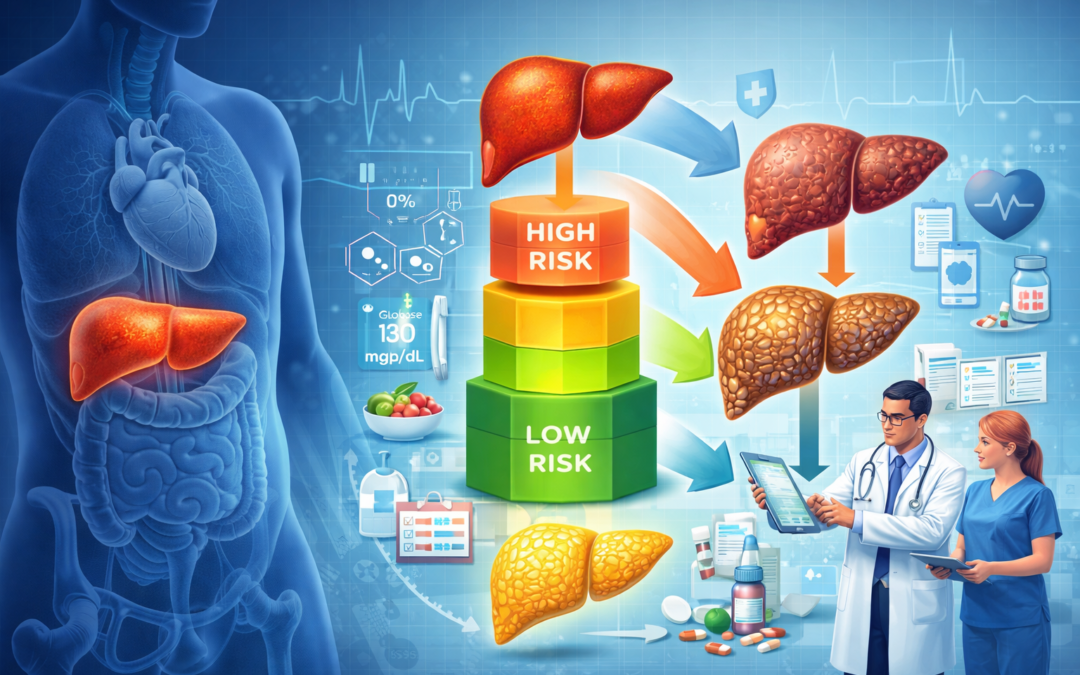
The newly proposed clinical care pathway for metabolic dysfunction-associated steatotic liver disease (MASLD) represents an important step toward standardizing the evaluation and management of one of the most prevalent chronic liver diseases globally. MASLD, closely linked to obesity, type 2 diabetes, and other cardiometabolic risk factors, is frequently managed in primary care and endocrinology settings, necessitating practical, scalable approaches to care.

Novo Nordisk has received US regulatory approval for Wegovy® HD (semaglutide 7.2 mg), representing a significant advancement in pharmacological obesity management. This higher-dose formulation of the GLP-1 receptor agonist semaglutide is designed to address unmet needs in patients who do not achieve adequate weight loss with existing therapies. The approval is supported by robust data from the phase 3 STEP UP clinical trial program, which evaluated the efficacy and safety of once-weekly semaglutide 7.2 mg in adults with obesity.






