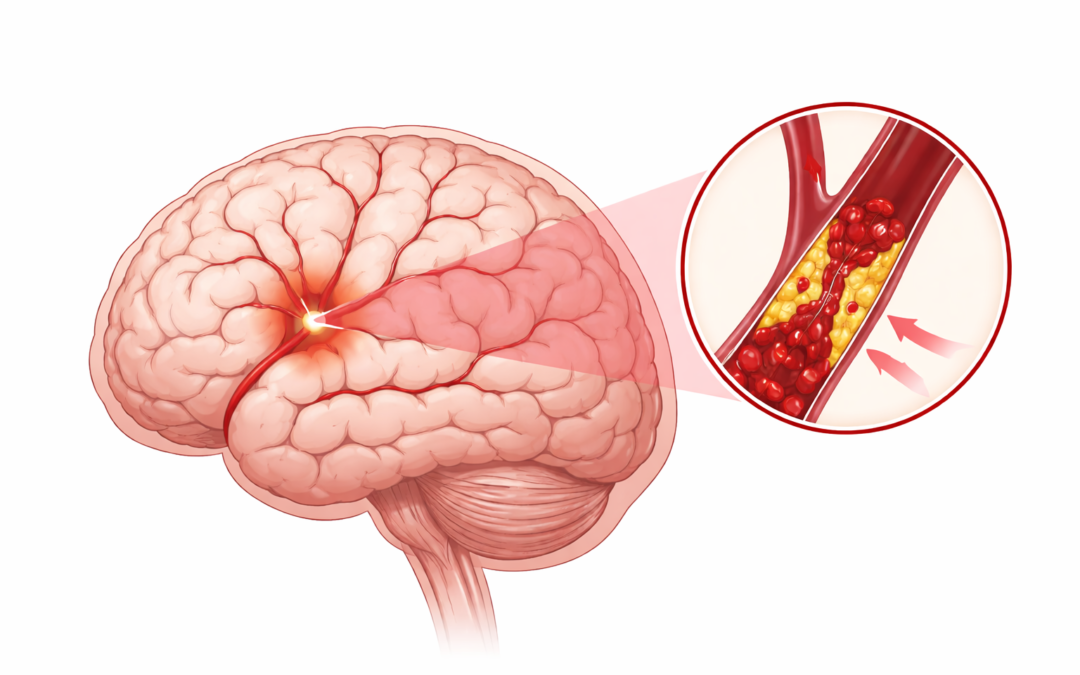
Acute ischemic stroke (AIS) remains a leading cause of disability and death worldwide. The 2026 Guideline for the Early Management of Patients With Acute Ischemic Stroke, from the American Heart Association/American Stroke Association (AHA/ASA), replaces the 2018 guideline and 2019 focused update. It incorporates evidence from randomized controlled trials, meta-analyses, and observational studies published through early 2025, addressing prehospital systems, emergency evaluation, reperfusion therapies, supportive care, in-hospital complications, and early secondary prevention for adults, with new guidance extending to select pediatric cases.
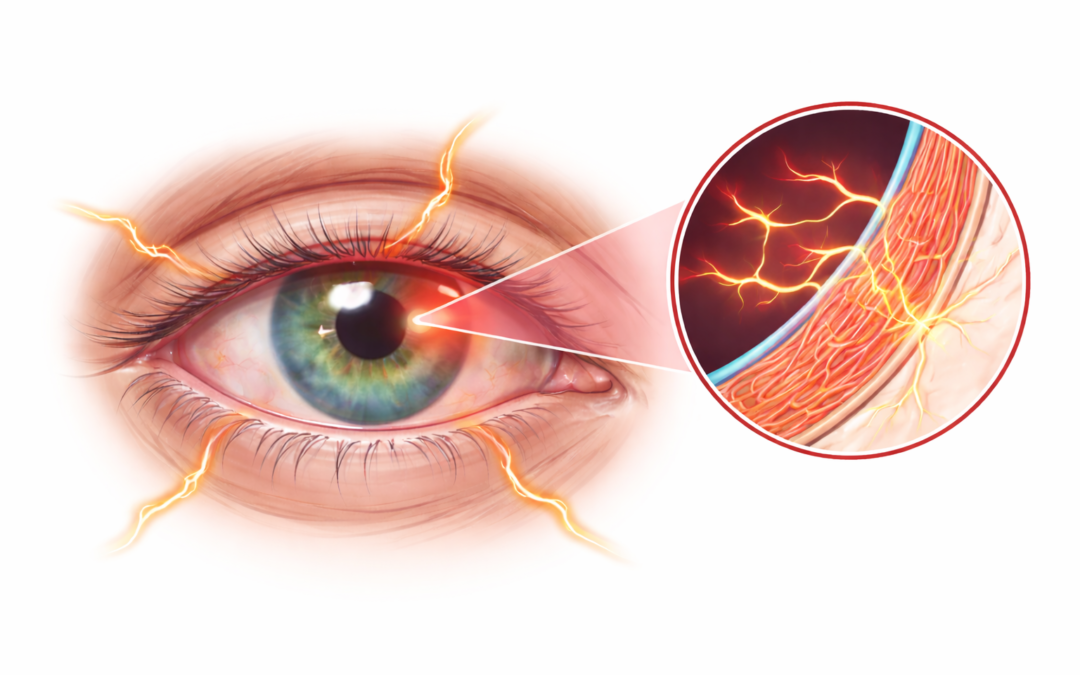
On January 23, 2026, OKYO Pharma Limited (Nasdaq: OKYO), a clinical-stage biopharmaceutical company focused on therapies for neuropathic corneal pain (NCP) and inflammatory eye diseases, announced that the U.S. Food and Drug Administration (FDA) has authorized a single-patient expanded access Investigational New Drug (IND 176297) application—commonly known as compassionate use—for urcosimod (0.05%). The IND was submitted by Pedram Hamrah, MD, at the University of South Florida, to treat a patient suffering from severe NCP who has exhausted available therapeutic options, with no FDA-approved treatments currently existing for this condition.

Non-communicable diseases (NCDs) account for 70% of global mortality, claiming over 38 million lives each year, with cardiovascular disease (CVD) as the leading contributor. While conventional risk factors like smoking, hypertension, and poor diet remain critical, emerging evidence highlights the escalating role of ubiquitous environmental risk factors (ERFs) in driving the rise of NCDs, particularly CVD. These interconnected anthropogenic exposures—air pollution, noise and light pollution, chemical and plastic contamination, water and soil pollution, and climate-related hazards—exert cumulative and compounding effects on cardiovascular health. They operate through shared pathophysiological mechanisms, including oxidative stress, systemic inflammation, autonomic nervous system imbalance, and endothelial dysfunction, amplifying overall risk beyond traditional factors.

Metformin remains a cornerstone first-line therapy for type 2 diabetes mellitus due to its efficacy, low cost, and favorable safety profile in patients with preserved renal function. However, its use is contraindicated in severe renal impairment (eGFR <30 mL/min/1.73 m²) owing to the risk of accumulation and metformin-associated lactic acidosis (MALA), with encephalopathy representing a rarer neurological manifestation. This report details a 44-year-old woman with type 2 diabetes, hypothyroidism, insomnia, hypertension, and end-stage diabetic nephropathy requiring maintenance hemodialysis three times weekly. One month after initiating immediate-release metformin 1000 mg/day at a primary care clinic, she presented to the emergency department with two days of dysarthria and generalized weakness. She was slightly somnolent but communicative, exhibited difficulty maintaining posture, gait instability, and no focal motor or sensory deficits.

On January 16, 2026, Novartis announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Therapy designation to its investigational therapy ianalumab (VAY736) for the treatment of Sjögren’s disease, the second most prevalent rheumatic autoimmune disease affecting approximately 0.25% of the population. Sjögren’s is a chronic, progressive, systemic condition characterized by mucosal dryness, fatigue, joint pain, and increased risk of lymphoma, often leading to significant quality-of-life impairment and frequently remaining undiagnosed or misdiagnosed. Currently, no approved targeted therapies exist, leaving patients with limited symptomatic management options.






