Insulin is the oldest of the currently available diabetes medications, has the most clinical experience. It is the most effective agent when used in adequate doses to decrease any level of HbA1c to the therapeutic goal. The updated ADA/EASD consensus recommends the use of GLP-1 receptor agonists or SGLT2 inhibitors with proven cardiovascular benefit in patients with type 2 diabetes and established ASCVD or high cardiovascular risk. Several studies have stated that initiation of insulin therapy is often delayed, leading to an increased risk of complications in patients with diabetes. The findings were presented at the EASD
Annual Meeting 2025, held from 15 -19 September 2025 in Vienna, Austria.

The REALIZE Study, a retrospective observational analysis, assessed the effectiveness and safety of early initiation of a fixed-dose combination (FDC) of dapagliflozin (SGLT-2 inhibitor) and sitagliptin (DPP-4 inhibitor) in 250 Indian patients with type 2 diabetes mellitus (T2DM) across five centers. Eligible patients, aged 18-59 years with a BMI ≥25 kg/m² and HbA1c between 7.0% and 10.5%, were prescribed the FDC for 112 ± 20 days.
Amgen announced on October 2, 2025, that the Phase 3 VESALIUS-CV trial successfully met its dual primary endpoints, establishing Repatha (evolocumab) as the first PCSK9 inhibitor to significantly reduce cardiovascular events in primary prevention settings. This randomized, double-blind, placebo-controlled study enrolled over 12,000 adults at high cardiovascular risk, including those with known atherosclerotic cardiovascular disease (ASCVD) or high-risk diabetes, but without prior myocardial infarction (MI) or stroke. Participants had elevated lipid levels (LDL-C ≥90 mg/dL, non-HDL-C ≥120 mg/dL, or apolipoprotein B ≥80 mg/dL) despite optimized lipid-lowering therapy, with approximately 85% on high- or moderate-intensity LDL-C reducing regimens like statins.
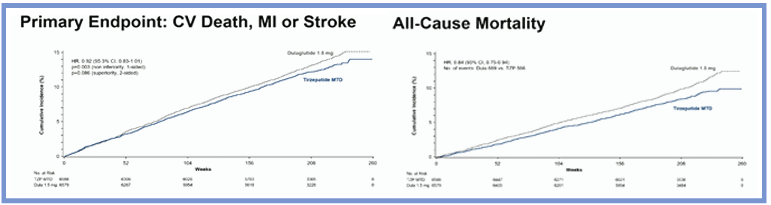
Tirzepatide, a dual GLP-1 and GIP receptor agonist, has been shown to improve glycaemic control and promote weight loss when compared with selective GLP-1 receptor agonists. Benefits of tirzepatide have also been observed for atherogenic lipoproteins, blood pressure, high sensitivity C-reactive protein and kidney function in comparison with selective GLP-1 receptor agonists or basal insulins. Stephen Nicholls assessed the cardiovascular outcomes in participants on tirzepatide versus dulaglutide in the SURPASS-CVOT trial. The findings were presented at the EASD Annual Meeting 2025, held th st from 15- 19 September 2025 in Vienna, Austria.
Dilated cardiomyopathy (DCM) is a leading cause of heart failure (HF), with left ventricular reverse remodeling (LVRR) linked to better outcomes, including reduced hospitalizations and mortality. While guideline-directed medical therapy (GDMT) promotes LVRR in HF with reduced ejection fraction (HFrEF), the role of ivabradine—a selective If-channel inhibitor recommended for HFrEF patients in sinus rhythm with resting heart rate (HR) ≥75 bpm despite GDMT—remains underexplored in relation to achieved HR and LVRR. This retrospective study at Severance Hospital (2012-2021) analyzed 255 patients with idiopathic non-ischemic DCM (NIDCM), defined by LVEF ≤35% and dilated left ventricle (LV), excluding ischemic, valvular, or other secondary etiologies via comprehensive diagnostics including cardiac MRI.






