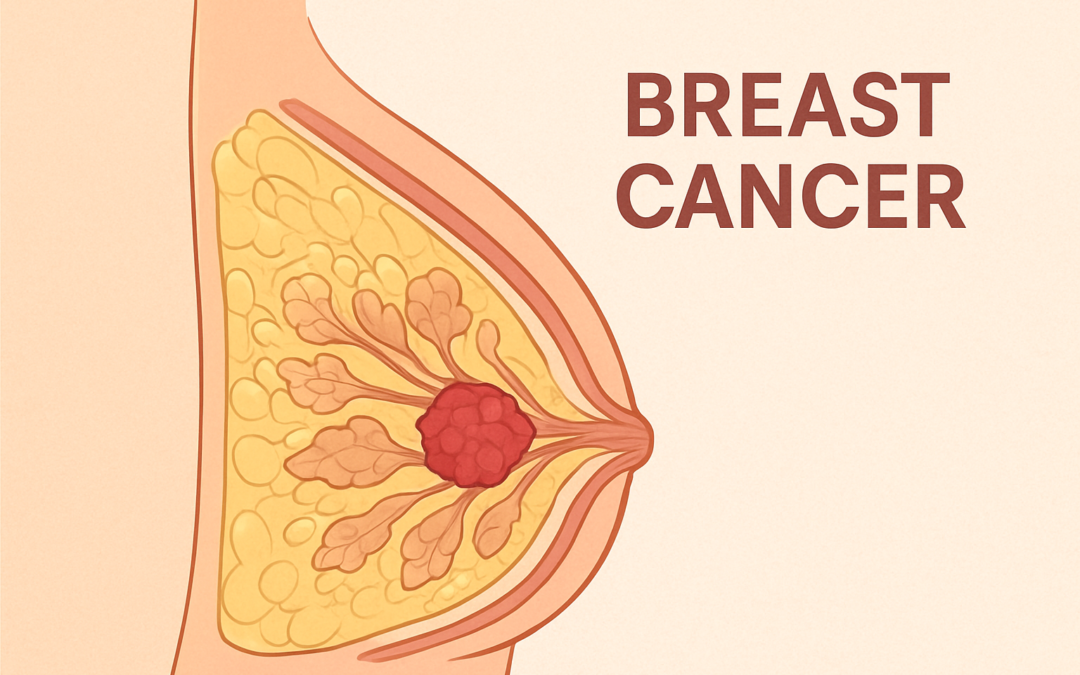
Eli Lilly and Company announced on September 25, 2025, that the U.S. Food and Drug Administration (FDA) has approved Inluriyo (imlunestrant), a 200 mg oral tablet, for the treatment of adults with estrogen receptor-positive (ER+), human epidermal growth factor receptor 2-negative (HER2-), ESR1-mutated advanced or metastatic breast cancer (MBC). This approval targets patients whose disease has progressed following at least one line of endocrine therapy (ET), addressing a critical need as approximately 50% of ER+, HER2- MBC patients develop ESR1 mutations after aromatase inhibitor exposure, leading to treatment resistance.
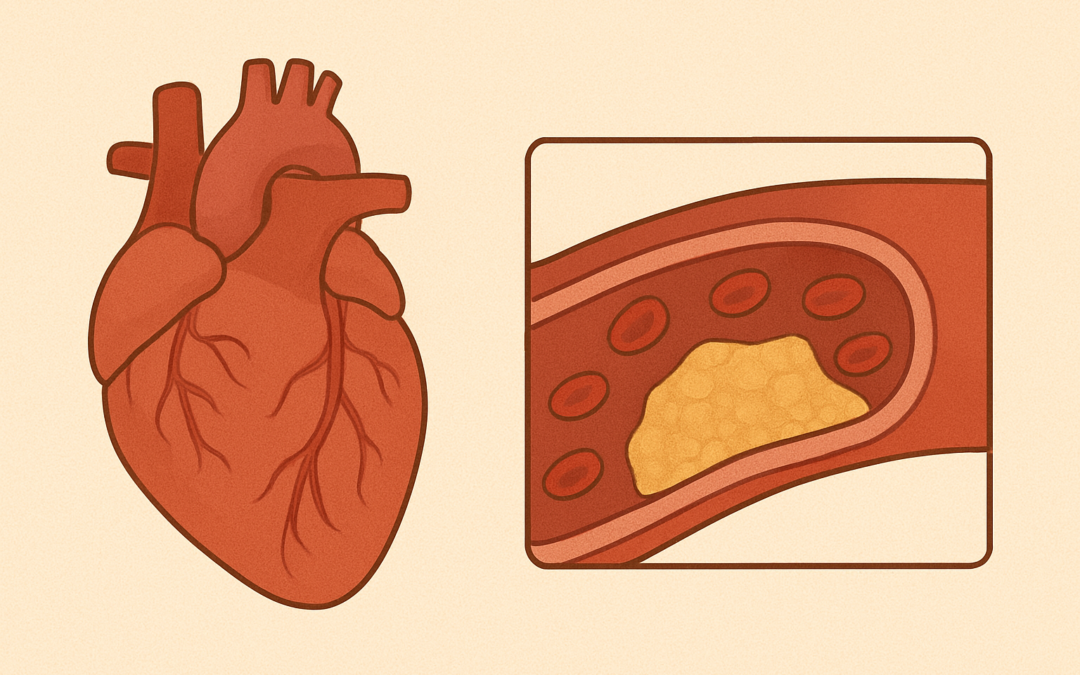
Atherosclerotic cardiovascular disease (ASCVD) poses a major threat to patients with type 2 diabetes, even without prior events. While aspirin is recommended for primary prevention in high-risk diabetic individuals, its benefits are offset by increased bleeding risks, particularly gastrointestinal (GI) bleeding. Clopidogrel, a P2Y12 inhibitor, has shown fewer GI events in secondary prevention trials, but its role in primary prevention remains unclear. This study aimed to compare the effectiveness and safety of clopidogrel versus aspirin in high- and very high-risk diabetic patients without ASCVD, using real-world data from the Korean National Health Insurance Service–National Sample Cohort (NHIS-NSC) from 2010-2019.
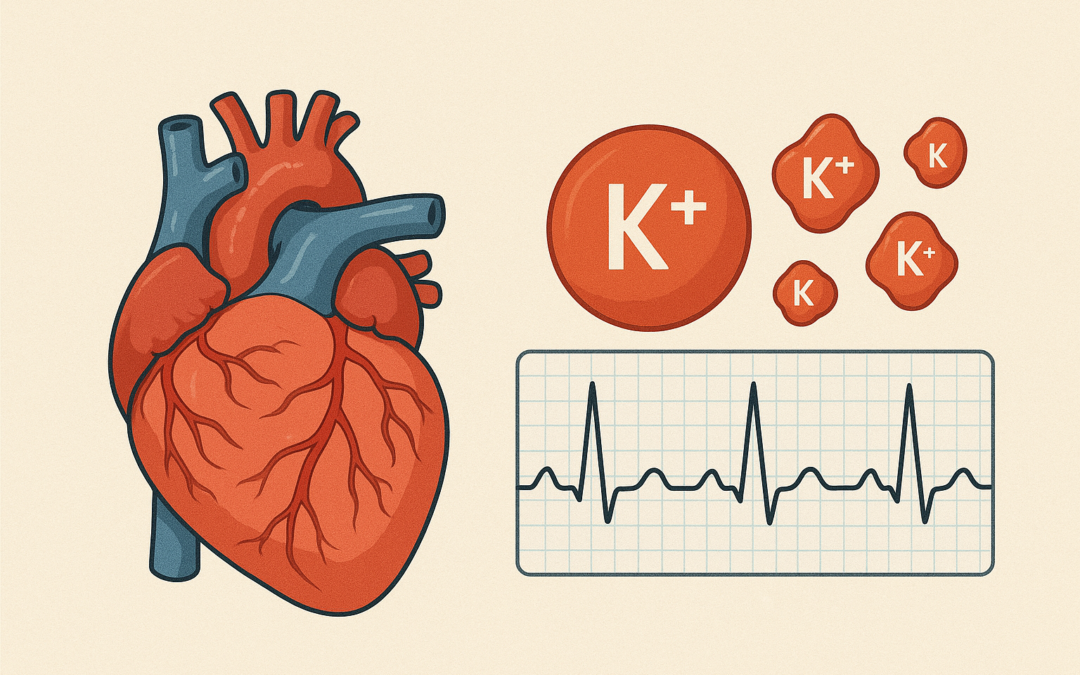
Heart failure with reduced ejection fraction (HFrEF) is a significant health issue, with sacubitril/valsartan (an angiotensin receptor neprilysin inhibitor) improving outcomes but potentially causing hyperkalemia due to aldosterone suppression and interactions with other therapies. This study evaluated potassium levels and hyperkalemia prevalence in HFrEF patients post-initiation, comparing pre- and post-treatment rates in a real-world Saudi cohort to inform monitoring strategies.

On September 25, 2025, the U.S. Food and Drug Administration (FDA) announced the approval of leucovorin calcium tablets for the treatment of cerebral folate deficiency (CFD), a neurological disorder characterized by impaired folate transport into the brain, resulting in developmental delays, autistic features (e.g., social communication challenges, sensory processing issues, repetitive behaviors), seizures, and motor coordination difficulties.

On September 22, 2025, the U.S. Food and Drug Administration (FDA) announced it is initiating a label change for acetaminophen, commonly known as Tylenol, to reflect evidence suggesting a potential association between its use during pregnancy and increased risks of autism spectrum disorder (ASD) and attention-deficit/hyperactivity disorder (ADHD) in offspring.






