On September 19, 2025, the U.S. Food and Drug Administration (FDA) approved Merck’s KEYTRUDA QLEX™ (pembrolizumab and berahyaluronidase alfa-pmph) injection for subcutaneous use in adults across 38 solid tumor indications previously approved for KEYTRUDA® (pembrolizumab), marking it as the first subcutaneously administered immune checkpoint inhibitor.
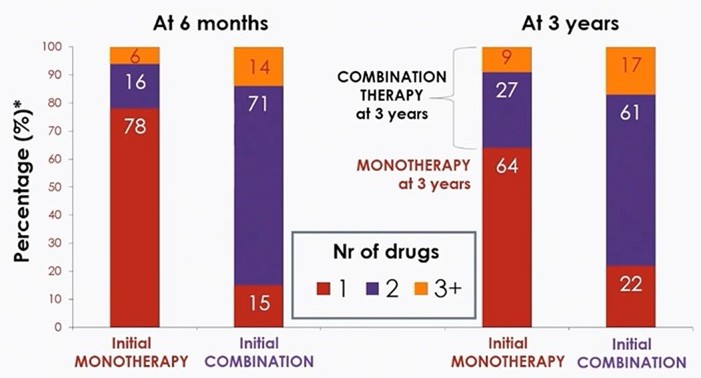
Hypertension remains a leading cause of cardiovascular morbidity and mortality worldwide, with suboptimal control and poor medication adherence posing significant challenges, particularly for cardiometabolic patients in 2025. Single-pill combinations (SPCs), which integrate multiple antihypertensive drugs into a single dose, have emerged as a transformative approach to address these issues. By simplifying treatment regimens, SPCs significantly improve patient adherence compared to multi-pill regimens, which often lead to missed doses and therapeutic failure.

Heart failure with preserved ejection fraction (HFpEF) represents a significant clinical challenge due to limited effective pharmacological treatments. Sodium-glucose co-transporter 2 (SGLT2) inhibitors, such as empagliflozin and dapagliflozin, have emerged as a novel therapeutic approach. This systematic review synthesizes evidence from 10 randomized controlled trials (RCTs) involving diverse HFpEF patient populations, including those with comorbidities like diabetes and chronic obstructive pulmonary disease.
Johnson & Johnson’s Innovative Medicine division marked a significant advancement in oncology on September 9, 2025, with the U.S. FDA approval of INLEXZO™ (gemcitabine intravesical system), formerly known as TAR-200. This novel therapy targets adult patients with Bacillus Calmette-Guérin (BCG)-unresponsive non-muscle invasive bladder cancer (NMIBC) characterized by carcinoma in situ (CIS), with or without papillary tumors—conditions affecting about 10% of NMIBC cases and leaving patients with few bladder-sparing alternatives post-BCG failure. INLEXZO™ addresses a critical unmet need in a field stagnant for over four decades, offering a potential paradigm shift by enabling extended local chemotherapy delivery without the morbidity of radical cystectomy, which carries a 3-8% postoperative mortality risk and profoundly impacts quality of life, particularly among older patients.
This secondary analysis of the ARCADIA trial, a randomized study comparing apixaban versus aspirin for secondary stroke prevention in patients with cryptogenic stroke and atrial cardiopathy, investigated the relationship between left ventricular (LV) systolic dysfunction and recurrent ischemic stroke, as well as the efficacy of apixaban in this context.






