Extensive-stage small cell lung cancer (ES-SCLC) is an aggressive malignancy with a poor prognosis, where first-line induction therapy typically involves platinum-etoposide chemotherapy combined with immunotherapy like atezolizumab, followed by maintenance to delay progression. Despite advances, relapse rates remain high, underscoring the need for effective maintenance strategies. On October 2, 2025, the U.S. Food and Drug Administration (FDA) approved lurbinectedin (Zepzelca), an alkylating agent that binds DNA guanine residues to disrupt the cell cycle and induce cancer cell death, in combination with atezolizumab (Tecentriq), a PD-L1 inhibitor, or its subcutaneous form (Tecentriq Hybreza), as first-line maintenance therapy for adult patients with ES-SCLC whose disease has not progressed after four cycles of induction therapy with atezolizumab, carboplatin, and etoposide. This approval, the first for a combination maintenance regimen in this setting, was based on the phase 3 IMforte trial (NCT05091567), a randomized, open-label, multicenter study involving 483 patients across 13 countries.

Introduction to Diabetes Distress:
Addressing the emotional side of diabetes and its management has received considerable attention in recent years. At the centre of most of these efforts is the concept of ‘diabetes distress’, a generic term that captures the primary sources and intensity of emotional distress associated with diabetes and its management over time. Karin Kanc discussed the recommendations for the clinical management of diabetes distress. The findings were presented at the EASD Annual th st Meeting 2025, held from 15 -19 September 2025 in Vienna, Austria.
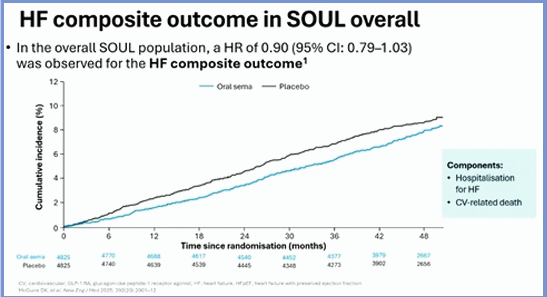
In clinical trials, once-weekly subcutaneous semaglutide reduced the risk of HF outcomes compared with placebo in individuals with HF (including those with preserved ejection fraction (HFpEF), obesity or chronic kidney disease). Data to benchmark results, in patients with cardiometabolic HFpEF semaglutide and tirzepatide showed >40% risk reduction for the composite of hospitalisation for HF or all-cause mortality compared with a placebo proxy. Rodica Pop-Busui aimed to assess the effects of oral semaglutide on heart failure outcomes in SOUL. The findings were presented at the EASD Annual th st Meeting 2025, held from 15 -19 September 2025 in Vienna, Austria.
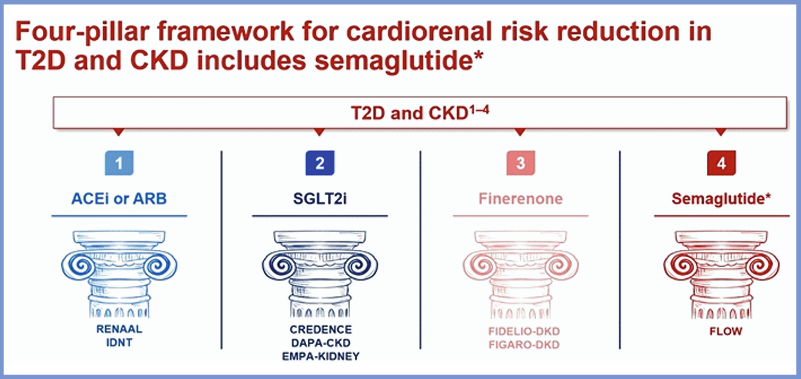
Diabetes is the leading cause of CKD and kidney failure. ~40% of people with T2D develop CKD. The risk of CV mortality increases with increased CKD staging, regardless of diabetes status. Paola Fioretto assessed the role of GLP-1RAs in the CKD and T2D nexus. The findings were presented at the th st EASD Annual Meeting 2025, held from 15-19 September 2025 in Vienna, Austria
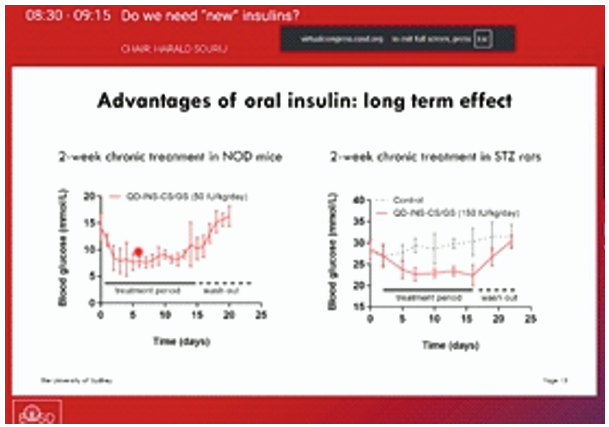
Insulin is highly sensitive to degradation from stomach acid and enzymes, but quantum dot (QD) conjugation can help make it insoluble across a wider pH range. While QD-insulin conjugation improved stability, QD-insulin alone was not effective in mice studies. The findings were presented at the EASD Annual Meeting 2025, held from 15 -19 September 2025 in Vienna, Austria.






